
424B5: Prospectus filed pursuant to Rule 424(b)(5)
Published on August 14, 2023
Filed Pursuant to Rule 424(b)(5)
Registration Statement No. 333-252715
PROSPECTUS SUPPLEMENT
(To Prospectus dated February 12, 2021)

Up to $50,000,000 of Shares of Common Stock
and 246,792 Shares of Common Stock
This prospectus supplement relates to the issuance and sale of up to $50,000,000 of shares of our common stock (“Purchase Shares”) that we may sell to Lincoln Park Capital Fund, LLC (“Lincoln Park”), from time to time pursuant to a purchase agreement, dated August 10, 2023 (the “Purchase Agreement”), that we have entered into with Lincoln Park, and an additional 246,792 shares of our common stock being issued to Lincoln Park as commitment shares under the Purchase Agreement.
This prospectus supplement also covers the resale of these shares by Lincoln Park to the public. See “Lincoln Park Transaction” for a description of the Purchase Agreement and additional information regarding Lincoln Park. Lincoln Park is an “underwriter” within the meaning of Section 2(a)(11) of the Securities Act of 1933, as amended (the “Securities Act”).
The purchase price for the Purchase Shares will be based upon formulas set forth in the Purchase Agreement depending on the type of purchase notice we submit to Lincoln Park from time to time. We will pay the expenses incurred in registering the shares of our common stock, including legal and accounting fees. See “Plan of Distribution” for more information.
Our common stock is listed on the Nasdaq Capital Market under the symbol “OCUP.” On August 10, 2023, the last reported sale price of our common stock on the Nasdaq Capital Market was $4.14 per share.
We are a “smaller reporting company” as defined under the federal securities laws and, as such, have elected to comply with certain reduced public company reporting requirements for this prospectus and the documents incorporated by reference herein and may elect to comply with reduced public company reporting requirements in future filings. See “Prospectus Supplement Summary-Implications of Being a Smaller Reporting Company.”
Investing in our securities involves a high degree of risk. See “Risk Factors” beginning on page S-6 of this prospectus supplement and in the documents incorporated by reference herein for a discussion of applicable information that should be considered in connection with an investment in our securities.
Neither the Securities and Exchange Commission nor any other regulatory body has approved or disapproved of these securities or passed upon the accuracy or adequacy of this prospectus supplement or the accompanying prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus supplement is August 11, 2023.
TABLE OF CONTENTS
Prospectus Supplement
| | | Page |
|
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
Prospectus
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
This prospectus supplement and the accompanying prospectus relate to the offering of our common stock. Before buying any of the common stock that we are offering, we urge you to carefully read this prospectus supplement and the accompanying prospectus, together with the information incorporated by reference as described under the headings “Where You Can Find Additional Information” and “Incorporation of Certain Information by Reference” in this prospectus supplement. These documents contain important information that you should consider when making your investment decision.
This prospectus supplement and the accompanying prospectus describes the terms of this offering of our common stock and also adds to and updates information contained in the documents incorporated by reference into this prospectus supplement and the accompanying prospectus. To the extent there is a conflict between the information contained in this prospectus supplement and the accompanying prospectus, on the one hand, and the information contained in any document incorporated by reference into this prospectus supplement and the accompanying prospectus that was filed with the SEC before the date of this prospectus supplement and the accompanying prospectus, on the other hand, you should rely on the information in this prospectus supplement and the accompanying prospectus. If any statement in one of these documents is inconsistent with a statement in another document having a later date (for example, a document incorporated by reference into this prospectus supplement and the accompanying prospectus) the statement in the document having the later date modifies or supersedes the earlier statement.
S-i
You should rely only on the information contained in or incorporated by reference in this prospectus supplement, the accompanying prospectus and in any free writing prospectus that we have authorized for use in connection with this offering. We have not, and Lincoln Park has not, authorized anyone to provide you with different information. If anyone provides you with different or inconsistent information, you should not rely on it. We are not, and Lincoln Park is not, making an offer to sell these securities in any jurisdiction where the offer or sale is not permitted. You should assume that the information appearing in this prospectus supplement and the accompanying prospectus, the documents incorporated by reference in this prospectus supplement, the accompanying prospectus, and in any free writing prospectus that we have authorized for use in connection with this offering, is accurate only as of the date of those respective documents. Our business, financial condition, results of operations and prospects may have changed since those dates. You should read this prospectus supplement and the accompanying prospectus, the documents incorporated by reference in this prospectus supplement, the accompanying prospectus, and any free writing prospectus that we have authorized for use in connection with this offering, in their entirety before making an investment decision.
“Ocuphire Pharma, Inc.” the “Ocuphire” logo and other trademarks, trade names or service marks of Ocuphire Pharma, Inc. appearing in this prospectus supplement and the accompanying prospectus are the property of Ocuphire Pharma, Inc. All other trademarks, trade names and service marks appearing in this prospectus supplement and the accompanying prospectus are the property of their respective owners. Solely for convenience, the trademarks and trade names in this prospectus may be referred to without the ® and ™ symbols, but such references should not be construed as any indicator that their respective owners will not assert their rights thereto.
S-ii
This summary highlights information contained elsewhere in this prospectus supplement and the accompanying prospectus. This summary is not complete and does not contain all of the information you should consider in making your investment decision. You should carefully read the entire prospectus supplement and accompanying prospectus, including the risks of investing in our securities discussed under the heading “Risk Factors” and under similar headings in the other documents that are incorporated by reference into this prospectus supplement and the accompanying prospectus. You should also carefully read the information incorporated by reference into this prospectus supplement and the accompanying prospectus, including our financial statements, and the exhibits to the registration statement of which this prospectus supplement and the accompanying prospectus are a part. Unless the context otherwise requires, the terms “Ocuphire” “the Company,” “we,” “us,” “our” and similar references in this prospectus refer to Ocuphire Pharma, Inc.
Overview
We are a clinical-stage ophthalmic biopharmaceutical company focused on developing novel therapies for the treatment of unmet needs of patients with retinal and refractive eye disorders.
APX3330
Our lead retinal product candidate, APX3330, is a first-in-class small-molecule inhibitor of Ref-1 (reduction oxidation effector factor-1 protein). Ref-1 is a regulator of transcriptor factors such as HIF-1α and NF-kB. Inhibiting REF-1 reduces levels of vascular endothelial growth factor (VEGF) and inflammatory cytokines which are known to play key roles in ocular angiogenesis and inflammation. Through inhibition of Ref-1, APX3330 normalizes the levels of VEGF to physiologic levels, unlike biologics that suppress VEGF below levels required for normal function. APX3330 is an oral tablet administered twice per day for the treatment of diabetic retinopathy (“DR”) and diabetic macular edema (“DME”).
DR affects approximately 10 million people with diabetes and is projected to impact 14.6 million Americans by 2050. DR is classified as Non-Proliferative Diabetic Retinopathy (“NPDR”), the early stage of the disease in which symptoms may be mild or nonexistent or Proliferative Diabetic Retinopathy (“PDR”) which is the more advanced stage of diabetic eye disease that can be highly symptomatic with loss of vision. Approximately 80% of the DR patients have NPDR that will progress to PDR if left untreated. Despite the risk for visual loss associated with this disease, over 90% of NPDR patients currently receive no course of treatment apart from observation by their eye care specialist until they develop sight-threatening complications. This is due to the burdensome and frequent eye injections required with currently approved therapies for this disease. APX3330 as an oral tablet has the potential to be an early, non-invasive treatment for the 8 million NPDR patients in the US.
In January 2023, we reported top-line efficacy and safety results from the ZETA-1 Phase 2 trial conducted in 103 subjects (51 treated with 600 mg daily dose of APX3330) in DR, including moderately severe NPDR and mild PDR, as well as patients with DME without loss of central vision. Although administration of APX3330 daily did not meet the study’s primary endpoint of a statistically significant percentage of patients with a ≥ 2-step improvement in Early Treatment of Diabetic Retinopathy Study (“ETDRS”) diabetic retinopathy severity scale (“DRSS”) in the study eye at week 24 compared to placebo, statistical significance was achieved on a key pre-specified secondary endpoint of preventing clinically meaningful progression of diabetic retinopathy (defined by binocular 3 or more steps worsening on the DRSS scale, calculated as the sum of the change in DRSS in the study eye and fellow eye) after 24 weeks of treatment. Given the systemic delivery of APX3330, an endpoint that evaluates the effects on both eyes is a potential FDA registration endpoint for future trials. The binocular ≥3-step worsening in DRSS endpoint will be confirmed at an End-of-Phase 2 (“EOP2”) meeting with the FDA in the fourth quarter of 2023.
Prior to Ocuphire in-licensing the APX3330 product candidate, it had been studied by other sponsors in a total of 11 clinical trials (6 Phase 1 and 5 Phase 2) in a total of over 420 healthy volunteers or patients (with over 340 APX3330-treated) for inflammatory (hepatic) and oncology indications, and had demonstrated evidence of target engagement, consistent pharmacokinetics, durability, and favorable safety and tolerability. Treatment-related adverse events were uncommon, and most were mild in severity. No clinically significant changes were observed in liver, kidney, or heart function. There were no treatment-related effects on hematologic or blood chemistry evaluations. APX3330 demonstrated favorable safety and tolerability in the ZETA-1 trial, consistent with the safety data from the prior 11 clinical trials.
S-1
We also in-licensed APX2009 and APX2014, which are second-generation analogs of APX3330. The unique dual mechanism of action of these Ref-1 inhibitors of reducing angiogenesis and inflammation could potentially be beneficial in treating other retinal diseases such as age-related macular degeneration, and geographic atrophy. We are currently evaluating local delivery routes in addition to the systemic (oral) route as part of a possible pipeline expansion in retinal therapies.
Nyxol
In November 2022, we entered into a license and collaboration agreement (the “Nyxol License Agreement”) with FamyGen Life Sciences, Inc. (acquired by Viatris, Inc. (“Viatris”) in January 2023) pursuant to which we granted Viatris an exclusive license to develop, manufacture, import, export and commercialize our refractive product candidate phentolamine ophthalmic solution 0.75% (Nyxol® Eye Drops or “Nyxol”) for treating (a) reversal of mydriasis, (b) night vision disturbances or dim light vision, and (c) presbyopia, and (ii) Nyxol and low dose pilocarpine for treating presbyopia (together, the “Nyxol Products”) worldwide except for certain countries and jurisdictions in Asia (the “Viatris Territory”).
Under the terms of the Nyxol License Agreement, Ocuphire, in partnership with Viatris, will develop the Nyxol Products in the United States. Viatris will reimburse us for budgeted costs related to the development of the Nyxol Products through FDA approval. Viatris will be responsible for developing the Nyxol Products in countries and jurisdictions in the Viatris Territory outside of the United States.
Nyxol is a once-daily eye drop formulation of phentolamine mesylate designed to reduce pupil diameter and improve visual acuity. Nyxol can potentially be used across multiple indications such as treatment of pharmacologically-induced mydriasis (“RM”) (dilation of the pupil), presbyopia (age-related blurry near vision) and dim light or night vision disturbances (“DLD”) (halos, glares and starbursts). Our management believes these multiple indications potentially represent a significant market opportunity. Nyxol has been studied in a total of 12 clinical trials (3 Phase 1, 5 Phase 2 and 4 Phase 3) in a total of over 1100 patients (with over 650 Nyxol-treated) and has demonstrated promising clinical data across the three targeted refractive indications.
We submitted a new drug application (“NDA”) to the U.S. Food and Drug Administration (“FDA”) in November 2022 under the 505(b)(2) pathway for Nyxol for RM with a Prescription Drug User Fee Act (PDUFA) goal date of September 28, 2023. Upon approval of Nyxol for RM, as per the Nyxol License Agreement, we will receive a $10 million milestone payment.
We reported positive topline data from multiple late-stage clinical trials for Nyxol in RM, presbyopia and DLD. We reported positive top-line data from Phase 3 trials in RM: MIRA-2 in March 2021, MIRA-3 in March 2022 and MIRA-4 in April 2022. We also reported positive top-line data from a Phase 2 trial of Nyxol for treatment of presbyopia, both as monotherapy and with low-dose pilocarpine (pilocarpine hydrochloride ophthalmic solution 0.4%, “LDP”) as adjunctive therapy (VEGA-1). We reported top-line data from a Phase 3 trial in DLD in May 2022 (LYNX-1). Funded by our partner, Vitaris, the first phase 3 registration trial of Nyxol for the treatment of presbyopia (VEGA-2), as monotherapy and with LDP as adjunctive therapy, was started in late December 2022, and topline results from this trial are expected in Q4 2023. Also funded by our partner Viatris, registration trials are planned for presbyopia and DLD as well as a supportive long-term safety trial for chronic use of Nyxol in refractive indications.
Corporate Information
Our principal executive offices are located at 37000 Grand River Avenue, Suite 120, Farmington Hills, MI 48335. Our telephone number is (248) 957-9024. Our website address is www.ocuphire.com. The information contained in, or accessible through, our website does not constitute part of this prospectus, and should not be relied on in determining whether to make an investment decision. The inclusion of our website address in this prospectus is an inactive textual reference only.
Implications of Being a Smaller Reporting Company
We are a “smaller reporting company” under federal securities laws. For as long as we continue to be a smaller reporting company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies, including, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements. We may continue to be a smaller reporting company if either (i) the market value of our stock held by non-affiliates is less than $250 million or (ii) our annual revenue was less than $100 million
S-2
during the most recently completed fiscal year and the market value of our stock held by non-affiliates is less than $700 million. As long as we remain a smaller reporting company and non-accelerated filer, we are exempt from the attestation requirement in the assessment of our internal control over financial reporting by our independent auditors pursuant to section 404(b) of the Sarbanes-Oxley Act of 2002 (the “Sarbanes-Oxley Act”) but are required to make our own internal assessment of the effectiveness of our internal controls over financial reporting.
S-3
The Offering
Common stock to be offered
246,792 shares of our common stock being issued to Lincoln Park as consideration for its commitment to purchase shares of our common stock under the Purchase Agreement (the “Commitment Shares”). We will not receive any cash proceeds from the issuance of these Commitment Shares.
Up to $50,000,000 of shares of our common stock we may issue and sell to Lincoln Park from time to time over the next 30 months, at our sole discretion, in accordance with the Purchase Agreement.
Common stock to be outstanding immediately after this offering
33,332,123 shares, based on 21,008,036 shares outstanding as of August 9, 2023, assuming sale of 12,077,295 shares at a price of $4.14 per share, which was the closing price of our common stock on The Nasdaq Capital Market on August 10, 2023. The actual number of shares issued will vary depending on the sales prices under this offering, but will not be greater than 4,195,058 shares, representing 19.99% of the shares of our common stock outstanding on the date of the Purchase Agreement (the “Exchange Cap”), unless, in accordance with the rules of The Nasdaq Stock Market LLC, unless we obtain stockholder approval of the issuance of shares of our common stock under the Purchase Agreement in excess of the Exchange Cap, or the average price of all applicable sales of our common stock to Lincoln Park under the Purchase Agreement is equal to or greater than the $4.399 (which represents the lower of (A) the official closing price of the Company’s Common Stock on Nasdaq on the date of the Purchase Agreement and (B) the average official closing price of the Company’s Common Stock on Nasdaq for the five consecutive trading days ending on the date of the Purchase Agreement, as adjusted to take into account the issuance of the Commitment Shares to Lincoln Park for non-cash consideration), such that the Exchange Cap will not apply under applicable Nasdaq listing rules to limit issuances and sales of Common Stock to Lincoln Park pursuant to the Purchase Agreement. The Purchase Agreement also provides that Lincoln Park will not purchase or acquire any shares of common stock under the Purchase Agreement which, when aggregated with all other shares of common stock beneficially owned by Lincoln Park to beneficially own more than 4.99% of our outstanding shares of common stock (the “Beneficial Ownership Cap”), which Lincoln Park may, in its sole discretion, increase to up to 9.99% of our outstanding shares of common stock by delivering written notice thereof to us, which shall not be effective until the 61st day after such written notice is delivered to us.
Use of proceeds
We may receive up to $50,000,000 in aggregate gross proceeds under the Purchase Agreement from any sales we make to Lincoln Park pursuant to the Purchase Agreement. We intend to use the net proceeds from this offering, if any, for clinical development of our product candidates and for
S-4
working capital and general corporate purposes. We do not currently have specific plans or commitments with respect to the net proceeds from this offering and, accordingly, we are unable to quantify the allocations of such proceeds among various potential uses. See “Use of Proceeds” on page S-12 of this prospectus supplement.
Risk factors
You should read the section entitled “Risk Factors” beginning on page S-6 and the documents incorporated by reference in this prospectus for a discussion of factors to consider carefully before deciding to invest in our common stock.
Nasdaq Capital Market symbol
“OCUP”
The number of shares of our common stock to be outstanding after this offering is based on 20,985,784 shares of common stock outstanding as of June 30, 2023, adjusted to include the issuance of 246,792 Commitment Shares being issued to Lincoln Park and excludes:
• |
3,444,656 shares of common stock issuable upon the exercise of outstanding stock options with a weighted-average exercise price of $3.00 per share; |
• |
5,665,838 shares of common stock issuable upon the exercise of outstanding warrants with an exercise price of $4.48 per share; |
• |
1,538,461 shares of common stock issuable upon the exercise of outstanding warrants with an exercise price of $6.09 per share; |
• |
58,597 shares of common stock issuable upon the exercise of outstanding warrants with an exercise price of $38.40 per share; |
• |
282,008 unvested restricted stock units; and |
• |
1,040,766 shares available for future issuance under the Ocuphire 2020 Equity Incentive Plan and the Ocuphire 2021 Inducement Plan. |
S-5
Investing in our common stock involves a high degree of risk. You should carefully consider the risks described below, and those discussed under the section entitled “Risk Factors” contained in our 2022 Annual Report on Form 10-K and our 2023 Quarterly Reports on Form 10-Q, together with other information in this prospectus supplement and the accompanying prospectus, the information and documents incorporated by reference herein, and in any free writing prospectus that we have authorized for use in connection with this offering. The occurrence of any of the events or developments described below could materially and adversely affect our business, financial condition, results of operations and prospects. In such an event, the market price of our common stock could decline and you may lose all or part of your investment.
Risks Related to this Offering
The sale or issuance of our common stock to Lincoln Park may cause dilution and the sale of the shares of common stock acquired by Lincoln Park, or the perception that such sales may occur, could cause the price of our common stock to fall.
On August 10, 2023, we entered into the Purchase Agreement with Lincoln Park, pursuant to which Lincoln Park has committed to purchase up to $50,000,000 of our common stock. Upon the execution of the Purchase Agreement, taking place on the date of this prospectus supplement, we are issuing 246,792 Commitment Shares to Lincoln Park, which represents the fee for its commitment to purchase shares of our common stock under the Purchase Agreement. We have also paid Lincoln Park $40,000 as full and complete reimbursement of its expenses in connection with the offering. The remaining shares of our common stock that may be issued under the Purchase Agreement may be sold by us to Lincoln Park at our discretion from time to time over a 30-month period commencing after the satisfaction of certain conditions set forth in the Purchase Agreement, including that the registration statement, of which this prospectus supplement and the accompanying prospectus form a part, is effective. The purchase price for the shares that we may sell to Lincoln Park under the Purchase Agreement will fluctuate based on the price of our common stock. Depending on market liquidity at the time, sales of such shares may cause the trading price of our common stock to fall.
We generally have the right to control the timing and amount of any future sales of our shares to Lincoln Park. Additional sales of our common stock, if any, to Lincoln Park will depend upon market conditions and other factors to be determined by us. We may ultimately decide to sell to Lincoln Park all, some or none of the additional shares of our common stock that may be available for us to sell pursuant to the Purchase Agreement. If and when we do sell shares to Lincoln Park, after Lincoln Park has acquired the shares, Lincoln Park may resell all, some or none of those shares at any time or from time to time in its discretion. Therefore, sales to Lincoln Park by us could result in substantial dilution to the interests of other holders of our common stock.
Additionally, the sale of a substantial number of shares of our common stock to Lincoln Park, or the anticipation of such sales, could make it more difficult for us to sell equity or equity-related securities in the future at a time and at a price that we might otherwise wish to effect sales.
A substantial number of shares of common stock may be sold in the market as a result of this offering, which may depress the market price for our common stock.
Sales of a substantial number of shares of our common stock in the public market as a result of this offering could cause the market price of our common stock to decline. A substantial majority of the outstanding shares of our common stock are, and the shares of common stock offered hereby will be, freely tradable without restriction or further registration under the Securities Act, unless these shares are owned or purchased by “affiliates” as that term is defined in Rule 144 under the Securities Act.
We have broad discretion to determine how to use the funds raised in this offering, if any, and may use them in ways that may not enhance our operating results or the price of our common stock.
Our management will have broad discretion over the use of net proceeds from this offering, if any, and we could spend the net proceeds from this offering in ways our stockholders may not agree with or that do not yield a favorable return, if at all. We currently expect to use the net proceeds from this offering, if any, for clinical development of our product candidates and for working capital and general corporate purposes, including costs and expenses associated with being a public company. However, our use of these net proceeds may differ substantially from our current plans.
S-6
If we do not invest or apply the net proceeds of this offering in ways that improve our operating results, we may fail to achieve expected financial results, which could cause our stock price to decline. Pending use, we may invest any net proceeds from this offering in a manner that does not produce income or loses value. See “Use of Proceeds” beginning on page S-12 of this prospectus supplement.
We may not be able to access sufficient funds under the Purchase Agreement when needed.
Our ability to sell shares to Lincoln Park and obtain funds under the Purchase Agreement is limited by the terms and conditions in the Purchase Agreement, including restrictions on the amounts we may sell to Lincoln Park at any one time, and a limitation on our ability to sell shares to Lincoln Park to the extent that it would cause Lincoln Park to beneficially own more than 4,195,058 shares in total under the Purchase Agreement, which is equal to 19.99% of the shares of common stock outstanding on the date of the Purchase Agreement, unless we obtain stockholder approval or the average price of such sales is equal to or greater than the minimum price of $4.399 as determined under Nasdaq rules. In addition, the Purchase Agreement also provides that Lincoln Park will not purchase or acquire any shares of common stock under the Purchase Agreement which, when aggregated with all other shares of common stock beneficially owned by Lincoln Park and its affiliates would result in the beneficial ownership of Lincoln Park and its affiliates of more than than 4.99% of our outstanding shares of common stock (the “Beneficial Ownership Cap”), which Lincoln Park may, in its sole discretion, increase to up to 9.99% of our outstanding shares of common stock by delivering written notice thereof to us, which shall not be effective until the 61st day after such written notice is delivered to us. In addition, any amounts we sell under the Purchase Agreement may not satisfy all of our funding needs, even if we are able and choose to sell and issue all of our common stock currently registered.
We may require additional financing to sustain our operations, without which we may not be able to continue operations, and the terms of subsequent financings may adversely impact our stockholders.
We may direct Lincoln Park to purchase up to $50,000,000 worth of shares of our common stock under our agreement over a 30-month period, in amounts up to 50,000 to 70,000 shares of our common stock depending on market prices, subject to a maximum limit of $1,000,000 per regular purchase, on any single business day.
Our ability to sell shares to Lincoln Park and obtain funds under the Purchase Agreement is limited by the terms and conditions in the Purchase Agreement, including restrictions on the amounts we may sell to Lincoln Park at any one time, and a limitation on our ability to sell shares to Lincoln Park to the extent that it would cause Lincoln Park to beneficially own more than 4.99% of our outstanding shares of common stock (the “Beneficial Ownership Cap”), which Lincoln Park may, in its sole discretion, increase to up to 9.99% of our outstanding shares of common stock by delivering written notice thereof to us, which shall not be effective until the 61st day after such written notice is delivered to us. Additionally, we are prohibited under applicable Nasdaq listing rules from issuing to Lincoln Park more than 4,195,058 shares of common stock under the Purchase Agreement (including Purchase Shares and Commitment Shares), such maximum number of shares representing 19.99% of the outstanding shares of our common stock immediately prior to the execution of the Purchase Agreement (such maximum share issuance limitation subject to certain reductions and pro rata adjustments as set forth in the Purchase Agreement, the “Exchange Cap”), unless (i) we first obtain stockholder approval in accordance with applicable Nasdaq listing rules to issue shares in excess of the Exchange Cap to Lincoln Park under the Purchase Agreement or (ii) the average price paid by Lincoln Park for all shares of common stock issued by us under the Purchase Agreement is equal to or greater than $4.399, such that the Exchange Cap will not apply under applicable Nasdaq listing rules to limit issuances and sales of our common stock to Lincoln Park pursuant to the Purchase Agreement. Therefore, we may not in the future have access to the full amount available to us under the Purchase Agreement, depending on the price of our common stock. In addition, any amounts we sell under the Purchase Agreement may not satisfy all of our funding needs, even if we are able and choose to sell and issue all of our common stock currently registered.
The extent to which we rely on Lincoln Park as a source of funding will depend on a number of factors including the prevailing market price of our common stock and the extent to which we are able to secure working capital from other sources. If obtaining sufficient funding from Lincoln Park were to prove unavailable or prohibitively dilutive, we may need to secure another source of funding in order to satisfy our future plan and working capital needs. Even if we sell all $50,000,000 of common stock under the Purchase Agreement to Lincoln Park, we may still need additional capital to finance our future plans and working capital needs, and we may have to raise funds through the issuance of equity or debt securities. Depending on the type and the terms of any financing we pursue, stockholders’ rights and the value of their investment in our common stock could be reduced. A financing could involve one or more types of securities including common stock, convertible debt or warrants to acquire common stock. These securities
S-7
could be issued at or below the then prevailing market price for our common stock. If the issuance of new securities results in diminished rights to holders of our common stock, the market price of our common stock could be negatively impacted. Should the financing we require to sustain our future plan and our working capital needs be unavailable or prohibitively expensive when we require it, the consequences could have a material adverse effect on our business, operating results, financial condition and prospects.
Our stock price and the trading volume of our stock may be volatile and you may not be able to resell shares of our common stock at or above the price you paid.
The trading price of our common stock could be highly volatile and could be subject to wide fluctuations in response to various factors, some of which are beyond our control. In particular, the trading prices for biopharmaceutical companies have been highly volatile as a result of supply chain disruptions and the ongoing COVID-19 pandemic. These factors include those described in this “Risk Factors” section and elsewhere in this prospectus supplement or the documents incorporated by reference herein, others such as:
• |
the announcement of new products or product enhancements by us or our competitors; |
• |
changes in our relationships with our licensors, licensees, or other strategic partners; |
• |
developments concerning intellectual property rights and regulatory approvals; |
• |
variations in our and our competitors’ results of operations; |
• |
the announcement of clinical trial results; |
• |
the announcement of potentially dilutive financings; |
• |
changes in earnings estimates or recommendations by securities analysts; |
• |
changes in the structure of healthcare payment systems; and |
• |
developments and market conditions in the pharmaceutical and biotechnology industries, including due to the COVID-19 pandemic. |
In addition, the stock markets in general, and the markets for biopharmaceutical stocks in particular, have experienced extreme volatility that may have been unrelated to the operating performance of the issuer. These broad market fluctuations may adversely affect the trading price or liquidity of our common stock.
S-8
This prospectus supplement and the documents we have filed with the SEC that are incorporated by reference contain “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended, or the Exchange Act. These statements relate to future events or to our future operating or financial performance and involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performances or achievements expressed or implied by the forward-looking statements. All statements other than statements of historical facts contained in this prospectus, including statements regarding our future results of operations and financial condition, business strategy and plans and objectives of management for future operations, are forward-looking statements. In some cases, forward-looking statements may be identified by words such as “anticipate,” “believe,” “continue,” “could,” “design,” “estimate,” “expect,” “intend,” “may,” “plan,” “potentially,” “predict,” “project,” “should,” “will” or the negative of these terms or other similar expressions.
We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. These forward-looking statements are subject to a number of known and unknown risks, uncertainties and assumptions, including risks described in the section titled “Risk Factors” and elsewhere in this prospectus and in our most recent Annual Report on Form 10-K and filed 2023 Quarterly Reports, as well as any amendments thereto reflected in subsequent filings with the SEC, which are incorporated by reference into this prospectus in their entirety, together with other information in this prospectus, the documents incorporated by reference and any free writing prospectus that we may authorize for use in connection with a specific offering. These risks include, among other things, that:
• |
we currently depend entirely on the success of Nyxol and APX3330, our only product candidates, and we may never complete clinical development of, receive marketing approval for, or successfully commercialize, Nyxol alone or as adjunctive therapy with low dose pilocarpine (LDP), APX3330, or other product candidates we may pursue in the future for any indication; |
• |
Viatris has exclusive rights to commercialize our Nyxol products in key global markets and Viatris’ failure to timely develop or commercialize these products would have a material adverse effect on our business and operating results; |
• |
the results of previous clinical trials may not be predictive of future results, and the results of our current and planned clinical trials may not satisfy the requirements of the FDA or non-U.S. regulatory authorities; |
• |
changes in regulatory requirements or FDA guidance, or unanticipated events during our clinical trials, may result in changes to clinical trial protocols or additional clinical trial requirements, which could result in increased costs to us or delays in our development timelines; |
• |
we expect to incur losses for the foreseeable future and may never achieve or maintain profitability. |
• |
adverse global economic conditions could have a negative effect on our business results of operations and financial condition and liquidity; |
• |
raising additional capital may cause dilution to our stockholders, restrict our operations, or require us to relinquish rights to our technologies or product candidates; |
• |
even if we receive marketing approval for our product candidates in the United States, we may never receive regulatory approval to market such product candidates outside of the United States; |
• |
our employees or our representatives may engage in misconduct or other improper activities, including violating applicable regulatory standards and requirements or engaging in insider trading, which could significantly harm our business; |
• |
we face substantial competition, which may result in others discovering, developing, or commercializing products before or more successfully than we do; |
• |
we lack experience in commercializing products, which may have an adverse effect on our business; |
S-9
• |
if we are unable to establish sales and marketing capabilities or enter into agreements with third parties to sell, market, and distribute APX3330, if approved, we may not be successful in commercializing APX3330 if and when it is approved; |
• |
product liability lawsuits against us, or our suppliers and manufacturers, could cause us to incur substantial liabilities and could limit commercialization of any product candidate that we may develop; |
• |
we are unable to control all aspects of our clinical trials due to our reliance on clinical research organizations, contract development and manufacturing organizations and other third parties that assist us in conducting clinical trials; |
• |
we are unable to control the supply, manufacture and testing of bulk drug substances and the formulation, testing and packaging of preclinical and clinical drug supplies of our product candidates, and will be unable to control these elements at the commercial stage, due to our reliance on third-party manufacturers and analytical facilities; |
• |
if we are not able to establish new collaborations for APX3330 on commercially reasonable terms, we may have to alter our development, manufacturing, and commercialization plans; |
• |
if we are unable to obtain and maintain sufficient patent protection for our product candidates, our competitors could develop and commercialize products or technology similar or identical to ours, which would adversely affect our ability to successfully commercialize any product candidates we may develop, our business, results of operations, financial condition and prospects; |
• |
if we do not obtain protection under the Hatch-Waxman Act and similar foreign legislation by extending the patent terms and obtaining data exclusivity for our product candidate, our business may be materially harmed; |
• |
we may not be able to protect or practice our intellectual property rights throughout the world; |
• |
obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment, and other requirements imposed by governmental agencies, and our patent protection could be reduced or eliminated for noncompliance with these requirements; |
• |
we depend on intellectual property sublicensed from Apexian Pharmaceuticals, Inc. (“Apexian”) for our APX3330 product candidate under development and our additional pipeline candidates, and the termination of, or reduction or loss of rights under, this sublicense would harm our business; |
• |
we are dependent on our key personnel, and if we are not successful in attracting and retaining highly qualified personnel, we may not be able to successfully implement our business strategy; |
• |
we will need to develop and expand our company and may encounter difficulties in managing this development and expansion, which could disrupt our operations; |
• |
our insurance policies are expensive and protect only from some business risk, which leaves us exposed to significant uninsured liabilities; |
• |
environmental, social, and governance matters and any related reporting obligations may impact our business; |
• |
if we fail to comply with the continued listing standards of the Nasdaq Capital Market, our common stock could be delisted. If it is delisted, our common stock price and the liquidity of our common stock would be impacted; |
• |
the market price of our common stock may fluctuate significantly; |
• |
we may be subject to securities litigation, which is expensive and could divert management attention.; and |
• |
there is uncertainty regarding the use of proceeds, if any, from this offering. |
These risks are not exhaustive. Other sections of this prospectus may include additional factors that could harm our business and financial performance. Moreover, we operate in a very competitive and rapidly changing environment.
S-10
New risks and uncertainties emerge from time to time, and it is not possible for our management to predict all risks and uncertainties nor can we assess the impact of all such factors on our business or the extent to which any such factor, or combination of such factors, may cause actual results to differ from those contained in, or implied by, any forward-looking statements.
You should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. Except as required by law, we undertake no obligation to update publicly any forward-looking statements for any reason after the date of this prospectus or to conform these statements to actual results or to changes in our expectations.
In addition, “we believe” and “we expect” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this prospectus, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements.
You should read this prospectus supplement and the documents that we reference in this prospectus and have filed as exhibits to the registration statement of which this prospectus is a part with the understanding that our actual future results, levels of activity, performance and achievements may be different from what we expect. We qualify all of our forward-looking statements by these cautionary statements.
S-11
We will receive no proceeds from the sale of shares of common stock by Lincoln Park in this offering. We may receive up to $50,000,000 in aggregate gross proceeds under the Purchase Agreement from any sales we make to Lincoln Park pursuant to the Purchase Agreement after the date of this prospectus supplement over a 30-month period, assuming that we sell the full amount of our common stock that we have the right, but not the obligation, to sell to Lincoln Park under the Purchase Agreement. The aggregate gross proceeds do not include offering expenses of approximately $140,000 and a cash fee of $40,000 to Lincoln Park as Lincoln Park's full and complete expense reimbursement in connection with entering into the transaction, including any legal fees. Because we are not obligated to sell any shares of our common stock under the Purchase Agreement, other than the Commitment Shares (from which we will receive no proceeds), the actual total offering amount and proceeds to us, if any, are not determinable at this time. There can be no assurance that we will receive any proceeds under or fully utilize the Purchase Agreement. See “Plan of Distribution” elsewhere in this prospectus supplement for more information.
We intend to use the net proceeds from the sale of common stock offered by us hereunder, if any, for clinical development of our product candidates and for working capital and other general corporate purposes.
Our expected use of the net proceeds from the sale of common stock offered by us hereunder, if any, represents our intentions based upon our current plans and business conditions. As of the date of this prospectus supplement, we cannot predict with certainty all of the particular uses for the net proceeds to be received upon the completion of this offering or the amounts that we will actually spend on the uses set forth above. The amounts and timing of our actual expenditures and the extent of our preclinical, clinical and future development activities may vary significantly depending on numerous factors, including the progress of our development efforts, the status of and results from our ongoing and planned clinical trials, our ability to take advantage of expedited programs or to obtain regulatory approval for product candidates, the timing and costs associated with the manufacture and supply of product candidates for clinical development and any unforeseen cash needs. As a result, our management will retain broad discretion over the allocation of the net proceeds from this offering.
Pending the use of the net proceeds from the sale of common stock offered by us hereunder, if any, as described above, we intend to invest the net proceeds, if any, in a variety of capital preservation instruments, including short-term, interest-bearing obligations, investment-grade instruments, certificates of deposit or direct or guaranteed obligations of the U.S. government.
S-12
The sale of our common stock to Lincoln Park pursuant to the Purchase Agreement may have a dilutive impact on our stockholders. In addition, the lower our stock price is at the time we exercise our right to sell shares to Lincoln Park, the more shares of our common stock we will have to issue to Lincoln Park pursuant to the Purchase Agreement and our existing stockholders will experience greater dilution.
Our historical net tangible book value as of June 30, 2023 was $37.7 million, or $1.80 per share of our common stock. Our historical net tangible book value represents our total tangible assets less total liabilities. Historical net tangible book value per share is our historical net tangible book value divided by the number of shares of our common stock outstanding as of June 30, 2023.
After giving further effect to the assumed sale by us of 12,077,295 shares of our common stock to Lincoln Park pursuant to the Purchase Agreement at an assumed average sale price of $4.14 per share of our common stock, which was the last reported sale price of our common stock on Nasdaq on August 10, 2023, and the issuance of 246,792 shares of common stock to Lincoln Park as Commitment Shares and without giving effect to the Exchange Cap under the Purchase Agreement, and after deducting estimated aggregate offering expenses payable by us and the fair value of Commitment Shares, our as pro forma adjusted net tangible book value as of June 30, 2023 would have been approximately $87.5 million, or $2.63 per share of common stock. This represents an immediate increase in pro forma net tangible book value per share of $0.83 to our existing stockholders and an immediate dilution in pro forma net tangible book value of $1.51 per share to Lincoln Park.
The following table illustrates this calculation on a per share basis. The as adjusted information is illustrative only and will adjust based on the actual price at which shares in this offering are sold and therefore, could result in an increase or decrease in net tangible book value per share as a result of this offering.
Assumed offering price per share
|
| | | | $4.14 |
|
Net tangible book value per share as of June 30, 2023
|
| | $1.80 |
| | |
Increase in pro forma net tangible book value per share attributable to Lincoln Park
|
| | 0.83 |
| | |
Pro forma as adjusted net tangible book value per share as of June 30, 2023 after giving effect to this offering
|
| | | | 2.63 |
|
Dilution in pro forma net tangible book value per share to Lincoln Park
|
| | | |
$1.51
|
The above table and discussion are based on 20,985,784 shares of common stock outstanding as of June 30, 2023 and exclude all of the following:
• |
3,444,656 shares of common stock issuable upon the exercise of outstanding stock options with a weighted-average exercise price of $3.00 per share; |
• |
5,665,838 shares of common stock issuable upon the exercise of outstanding warrants with an exercise price of $4.48 per share; |
• |
1,538,461 shares of common stock issuable upon the exercise of outstanding warrants with an exercise price of $6.09 per share; |
• |
58,597 shares of common stock issuable upon the exercise of outstanding warrants with an exercise price of $38.40 per share; |
• |
282,008 unvested restricted stock awards; and |
• |
1,040,766 shares available for future issuance under the Ocuphire 2020 Equity Incentive Plan and the Ocuphire 2021 Inducement Plan. |
To the extent that options or warrants outstanding as of June 30, 2023 have been or are exercised, or other shares are issued, investors purchasing shares in this offering could experience further dilution. In addition, we may choose to raise additional capital due to market conditions or strategic considerations, even if we believe we have sufficient funds for our current or future operating plans. To the extent that additional capital is raised through the sale of equity or convertible debt securities, the issuance of these securities could result in further dilution to our stockholders.
S-13
General
On August 10, 2023, we entered into the Purchase Agreement with Lincoln Park. In connection with the Purchase Agreement, on August 10, 2023, we also entered into a registration rights agreement (the “Registration Rights Agreement”) with Lincoln Park, pursuant to which we agreed to take specified actions to maintain the registration of the shares of our common stock subject to the offering described in this prospectus supplement and the accompanying prospectus. Pursuant to the terms of the Purchase Agreement, Lincoln Park has agreed to purchase from us up to $50,000,000 of our common stock (subject to certain limitations) from time to time during the 30-month term of the Purchase Agreement. Pursuant to the terms of the Purchase Agreement and Registration Rights Agreement, we have filed with the SEC this prospectus supplement regarding the sale under the Securities Act of the shares issuable to Lincoln Park under the Purchase Agreement. Pursuant to the terms of the Purchase Agreement, on the date of this prospectus supplement, we are issuing 246,792 Commitment Shares to Lincoln Park as consideration for its commitment to purchase shares of our common stock under the Purchase Agreement. We have also paid a cash fee of $40,000 to Lincoln Park as Lincoln Park’s full and complete expense reimbursement in connection with entering into the transaction, including any legal fees.
We may, from time to time and at our sole discretion, direct Lincoln Park to purchase shares of our common stock upon the satisfaction of certain conditions set forth in the Purchase Agreement at purchase prices per share as computed in accordance with the terms as set forth in the Purchase Agreement. We will control the timing and amount of any sales of our common stock to Lincoln Park, and Lincoln Park has no right to require us to sell any shares to it under the Purchase Agreement. Lincoln Park may not assign or transfer its rights and obligations under the Purchase Agreement.
Under applicable Nasdaq listing rules, we are prohibited from issuing to Lincoln Park more than the Exchange Cap of 4,195,058 shares of common stock under the Purchase Agreement (including Purchase Shares and Commitment Shares), representing 19.99% of the outstanding shares of our common stock immediately prior to the execution of the Purchase Agreement (subject to certain reductions and pro rata adjustments as set forth in the Purchase Agreement), unless (i) we first obtain stockholder approval in accordance with applicable Nasdaq listing rules to issue shares in excess of the Exchange Cap to Lincoln Park under the Purchase Agreement or (ii) the average price paid by Lincoln Park for all shares of common stock issued by us under the Purchase Agreement is equal to or greater than $4.399 (which represents the lower of (A) the official closing price of our common stock on Nasdaq on the date of the Purchase Agreement and (B) the average official closing price of our common stock on Nasdaq for the five consecutive trading days ending on the date of the Purchase Agreement, as adjusted to take into account our issuance of the Commitment Shares to Lincoln Park for non-cash consideration), such that the Exchange Cap will not apply under applicable Nasdaq listing rules to limit issuances and sales of our common stock to Lincoln Park pursuant to the Purchase Agreement. In any event, the Purchase Agreement specifically provides that we may not issue or sell any shares of our common stock under the Purchase Agreement if such issuance or sale would breach any applicable Nasdaq listing rules.
The Purchase Agreement also prohibits us from directing Lincoln Park to purchase any shares of our common stock if those shares of our common stock, when aggregated with all other shares of our common stock then beneficially owned by Lincoln Park and its affiliates (as calculated pursuant to Section 13(d) of the Securities Exchange Act of 1934, as amended, and Rule 13d-3 thereunder), would result in Lincoln Park having beneficial ownership of shares of our common stock in excess of the 4.99% Beneficial Ownership Cap, which Beneficial Ownership Cap Lincoln Park may, in its sole discretion, increase to up to 9.99% of our outstanding shares of common stock by delivering written notice thereof to us, which shall not be effective until the 61st day after such written notice is delivered to us.
Purchase of Shares under the Purchase Agreement
Regular Purchases
Under the Purchase Agreement, after the Commencement Date (as defined below), on any business day selected by us, we may direct Lincoln Park to purchase up to 50,000 shares of our common stock on such business day (or the purchase date), which we refer to as a Regular Purchase, provided that the Closing Sale Price of our common stock on the applicable Purchase Date is not below $0.25, provided, however, that (i) a Regular Purchase may be increased to up to 60,000 shares if the closing sale price of our common stock on Nasdaq is not below $5.00 on the
S-14
applicable purchase date and (ii) a Regular Purchase may be increased to up to 70,000 shares if the closing sale price of our common stock on Nasdaq is not below $7.50 on the applicable purchase date. Lincoln Park’s committed obligation under any single regular purchase, subject to certain exceptions, cannot exceed $1,000,000. We may direct Lincoln Park to purchase shares in Regular Purchases as often as every business day. The foregoing share amounts and per share prices will be adjusted for any reorganization, recapitalization, non-cash dividend, stock split, reverse stock split or other similar transaction occurring after the date of the Purchase Agreement.
The purchase price per share for each such Regular Purchase will be equal to the lesser of:
• |
the lowest sale price for our common stock on Nasdaq on the purchase date of such shares; and |
• |
the arithmetic average of the three (3) lowest closing sale prices for our common stock on Nasdaq during the ten (10) consecutive business days prior to the purchase date of such shares. |
Accelerated Purchases
We may also direct Lincoln Park, on any business day on which we have submitted a Regular Purchase notice for the maximum amount allowed for such Regular Purchase, to purchase an additional amount of our common stock, which we refer to as an Accelerated Purchase, of up to the lesser of:
• |
three (3) times the number of shares purchased pursuant to such Regular Purchase; or |
• |
30% of the aggregate shares of our common stock traded on Nasdaq during the period on the trading day immediately following the purchase date for such Regular Purchase (the “Accelerated Purchase Date”), beginning at the commencement of regular trading on Nasdaq (or such later time on such Accelerated Purchase Date as mutually agreed by us and Lincoln Park specified in the Accelerated Purchase notice for such Accelerated Purchase), and ending at the close of regular trading on Nasdaq on such Accelerated Purchase Date, or, if certain trading volume or market price thresholds specified in the Purchase Agreement are crossed prior to the close of regular trading on Nasdaq on the applicable Accelerated Purchase Date, ending at such earlier time that any one of such thresholds is crossed, which period of time on the applicable Accelerated Purchase Date (the “Accelerated Purchase Measurement Period”). |
The purchase price per share for each such Accelerated Purchase will be equal to 96.5% of the lower of:
• |
the closing sale price of our common stock on Nasdaq on the applicable Accelerated Purchase date; and |
• |
the volume-weighted average price of our common stock on Nasdaq during the applicable Accelerated Purchase Measurement Period on the applicable Accelerated Purchase date. |
Additional Accelerated Purchases
We may also direct Lincoln Park on any business day on which an Accelerated Purchase has been completed and all of the shares to be purchased thereunder have been delivered to Lincoln Park in accordance with the Purchase Agreement, to purchase an additional amount of our common stock, which we refer to as an Additional Accelerated Purchase, of up to the lesser of:
• |
three (3) times the number of shares purchased pursuant to the applicable corresponding Regular Purchase; and |
• |
30% of the aggregate shares of our common stock traded on Nasdaq during the period on the applicable Additional Accelerated Purchase Date beginning at the time mutually agreed by us and Lincoln Park and specified in the Additional Accelerated Purchase notice for such Additional Accelerated Purchase, and ending at the close of regular trading on Nasdaq on such Additional Accelerated Purchase Date, or, if certain trading volume or market price thresholds specified in the Purchase Agreement are crossed prior to the close of regular trading on Nasdaq on such date, ending at such earlier time that any one of such thresholds is crossed, which period of time on the applicable Additional Accelerated Purchase Date (the “Additional Accelerated Purchase Measurement Period”). |
We may, in our sole discretion, submit multiple Additional Accelerated Purchase notices to Lincoln Park on a single Accelerated Purchase date, provided that all prior Accelerated Purchases and Additional Accelerated Purchases (including those that have occurred earlier on the same day) have been completed and all of the shares to be purchased thereunder have been properly delivered to Lincoln Park in accordance with the Purchase Agreement.
S-15
The purchase price per share for each such Additional Accelerated Purchase will be equal to 96.5% of the lower of:
• |
the closing sale price of our common stock on Nasdaq on the applicable Additional Accelerated Purchase date; and |
• |
the volume-weighted average price of our common stock on Nasdaq during the applicable Additional Accelerated Purchase Measurement Period on the applicable Additional Accelerated Purchase Date. |
In the case of Regular Purchases, Accelerated Purchases, and Additional Purchases, the purchase price per share will be equitably adjusted for any reorganization, recapitalization, non-cash dividend, stock split, reverse stock split or other similar transaction occurring between the business days used to compute the purchase price. Other than as set forth above, there are no trading volume requirements or restrictions under the Purchase Agreement, and we will control the timing and amount of any sales of our common stock to Lincoln Park.
Suspension Events
Suspension events under the Purchase Agreement include the following:
• |
the effectiveness of the registration statement of which this prospectus supplement and accompanying prospectus form a part lapses for any reason (including, without limitation, the issuance of a stop order or similar order by the SEC), or such registration statement (or the prospectus forming a part thereof) is unavailable for the sale by us or the resale by Lincoln Park of our common stock under the Purchase Agreement, and such lapse or unavailability continues for a period of 10 consecutive business days or for more than an aggregate of 30 business days in any 365-day period, but excluding a lapse or unavailability where (i) we terminate the registration statement after Lincoln Park has confirmed in writing that all of the shares of our common stock covered thereby have been resold or (ii) we supersede the registration statement with a new registration statement, including, without limitation, when the prior registration statement is effectively replaced with a new registration statement covering the shares of our common stock covered by the Purchase Agreement (provided in the case of this clause (ii) that all of the shares of our common stock covered by the superseded or terminated registration statement that have not theretofore been sold to Lincoln Park are included in the superseding or new registration statement); |
• |
suspension by Nasdaq of our common stock from trading or the failure of the common stock to be listed on Nasdaq for a period of one business day; |
• |
the delisting of our common stock from Nasdaq Capital Market, provided, however, that the common stock is not immediately thereafter trading on the Nasdaq Global Market, the Nasdaq Global Select Market, the New York Stock Exchange, the NYSE American, the NYSE Arca, or the OTCQX or OTCQB operated by the OTC Markets Group, Inc. (or any nationally recognized successors thereto); |
• |
the failure for any reason by our transfer agent to issue (i) the Commitment Shares to Lincoln Park within two business days after the commencement date of the Purchase Agreement, or (ii) Purchase Shares to Lincoln Park within two business days after any Regular Purchase date, Accelerated Purchase Date or Additional Accelerated Purchase Date, as applicable, on which Lincoln Park is entitled to receive such shares; |
• |
if we breach any representation, warranty, covenant, or other term or condition contained in the Purchase Agreement or Registration Rights Agreement if such breach would reasonably be expected to have a Material Adverse Effect (as defined in the Purchase Agreement) and, except, in the case of a breach of a covenant that is reasonably curable, only if such breach continues for a period of at least five business days; |
• |
if any person commences a proceeding against the us pursuant to or within the meaning of any bankruptcy law; |
• |
if we, pursuant to or within the meaning of any bankruptcy law, (i) commence a voluntary case, (ii) consent to the entry of an order for relief against us in an involuntary case, (iii) consent to the appointment of a custodian of the Company or for all or substantially all of our property, or (iv) makes a general assignment for the benefit of our creditors or are generally unable to pay our debts as the same become due; |
• |
a court of competent jurisdiction enters an order or decree under any bankruptcy law that (i) is for relief against us in an involuntary case, (ii) appoints a custodian of the Company or for all or substantially all of our property, or (iii) orders the liquidation of us or any of our subsidiaries; |
S-16
• |
if, at any time, we are not eligible to transfer our common stock electronically as DWAC shares; or |
• |
if, at any time after the commencement date of the Purchase Agreement, the Exchange Cap is reached (to the extent such Exchange Cap is applicable) and the stockholder approval to issue in excess of such amount has not been obtained in accordance with the applicable rules of Nasdaq. |
In addition to any other rights and remedies under applicable law and the Purchase Agreement, so long as a suspension event has occurred and is continuing, or if any event which, after notice and/or lapse of time, would become a suspension event has occurred and is continuing, we shall not deliver to Lincoln Park any Regular Purchase notice, Accelerated Purchase notice or Additional Accelerated Purchase notice. Lincoln Park does not have the right to terminate the Purchase Agreement upon any of the suspension events set forth above, however, the Purchase Agreement will automatically terminate (i) if, pursuant to or within the meaning of any bankruptcy law, we commence a voluntary case or if any person commences a proceeding against us which is not discharged within 90 days, or a custodian is appointed for the Company or for substantially of all of our property, or we make a general assignment for the benefit of our creditors; (ii) we sell and Lincoln Park purchases the full amount of shares available under the Purchase Agreement; or (iii) the full amount of shares available under the Purchase Agreement has not been purchased by the end of the 30-month period commencing after the satisfaction of certain conditions set forth in the Purchase Agreement.
Our Termination Rights
We have the unconditional right, at any time, for any reason and without any payment or liability to us, to give one (1) business day notice to Lincoln Park to terminate the Purchase Agreement.
No Short-Selling or Hedging by Lincoln Park
Lincoln Park has represented and warranted that neither it nor any of its agents, representatives or affiliates has engaged in any direct or indirect short-selling or hedging of our common stock, and has agreed that neither it nor any of its agents, representatives or affiliates will engage in any direct or indirect short-selling or hedging of our common stock during any time prior to the termination of the Purchase Agreement.
Prohibitions on Variable Rate Transactions
Subject to specified exceptions included in the Purchase Agreement, we are limited in our ability to enter into specified variable rate transactions until the 30-month anniversary of the date of the Purchase Agreement, irrespective of any earlier termination of the Purchase Agreement. Such transactions include an equity line of credit, at-the-market offering, or other similar continuous offering in which we may offer or issue or sell our common stock or other securities that entitle the holder thereof to acquire shares of our common stock at a future determined price. However, we are permitted to enter into certain at-the-market offerings exclusively through a registered broker-dealer acting as agent of the Company pursuant to a written agreement between the Company and such registered broker-dealer, including our existing at-the-market offering.
Effect of Performance of the Purchase Agreement on our Stockholders
All shares registered in this offering that have been or may be issued or sold by us to Lincoln Park under the Purchase Agreement are expected to be freely tradable. Shares registered in this offering may be sold over a period of up to 30 months commencing on the date of this prospectus supplement. The sale by Lincoln Park of a significant amount of shares registered in this offering at any given time could cause the market price of our common stock to decline and to be highly volatile. Sales of our common stock to Lincoln Park, if any, will depend upon market conditions and other factors to be determined by us. We may ultimately decide to sell to Lincoln Park all, some or none of the additional shares of our common stock that may be available for us to sell pursuant to the Purchase Agreement. If and when we do sell shares to Lincoln Park, after Lincoln Park, has acquired the shares, Lincoln Park may resell all, some or none of those shares at any time or from time to time in its discretion. Therefore, sales to Lincoln Park by us under the Purchase Agreement may result in substantial dilution to the interests of other holders of our common stock. In addition, if we sell a substantial number of shares to Lincoln Park under the Purchase Agreement, or if investors expect that we will do so, the actual sales of shares or the mere existence of our arrangement with Lincoln Park may make it more difficult for us to sell equity or equity-related securities in the future at a time and at a price that we might otherwise wish to effect such sales. However, we have the right to control the timing and amount of any additional sales of our shares to Lincoln Park and the Purchase Agreement may be terminated by us at any time at our discretion without any cost to us.
S-17
Pursuant to the terms of the Purchase Agreement, we have the right, but not the obligation, to direct Lincoln Park to purchase up to $50,000,000 of our common stock, exclusive of the Initial Commitment Shares being issued to Lincoln Park as consideration for its commitment to purchase shares of our common stock under the Purchase Agreement and the Additional Commitment Shares that may be issued to Lincoln Park in connection with each purchase under the Purchase Agreement. The Purchase Agreement generally prohibits us from issuing or selling to Lincoln Park under the Purchase Agreement (i) shares of our common stock in excess of the Exchange Cap, unless we obtain stockholder approval to issue shares in excess of the Exchange Cap or the shares we desire to sell in excess of the Exchange Cap must be sold for an average price that equals or exceeds $4.399 per share (subject to adjustment for any reorganization, recapitalization, non-cash dividend, stock split, reverse stock split or other similar transaction that occurs on or after the date of the Purchase Agreement), such that the transactions contemplated by the Purchase Agreement are exempt from the Exchange Cap limitation under applicable Nasdaq rules and (ii) any shares of our common stock if those shares, when aggregated with all other shares of our common stock then beneficially owned by Lincoln Park and its affiliates, would exceed the Beneficial Ownership Limitation of our then issued and outstanding shares of common stock.
The following table sets forth the amount of gross proceeds we would receive from Lincoln Park from our sale of shares to Lincoln Park under the Purchase Agreement at varying purchase prices:
|
Assumed Average
Purchase Price
|
| |
Number of Registered
Shares to be Issued
if Full Purchase(1)
|
| |
Percentage of
Outstanding Shares
After Giving Effect to
the Issuance to
Lincoln Park(2)
|
| |
Gross Proceeds to Us
from the Sale
of Shares to Lincoln
Park Under the
Purchase Agreement
|
$2.00
|
| |
4,195,058(3)
|
| | 16.50% |
| | $8,390,116
|
$3.00
|
| |
4,195,058(3)
|
| | 16.50% |
| | $12,585,174 |
$4.14(4)
|
| |
4,195,058(3)
|
| | 16.50% |
| | $17,367,540 |
$5.00
|
| | 10,000,000 |
| | 32.02% |
| | $50,000,000 |
$6.00
|
| | 8,333,333 |
| | 28.19% |
| | $50,000,000 |
$7.00
|
| | 7,142,857 |
| | 25.17% |
| | $50,000,000 |
(1) |
Includes the total number of Purchase Shares which we would have sold under the Purchase Agreement at the corresponding assumed average purchase price set forth in the first column, up to the aggregate purchase price of $50,000,000, if available, while giving effect to the Exchange Cap and without regard for the limitation of 9.99% of our outstanding shares of common stock that Lincoln Park may beneficially own under the Purchase Agreement, and excludes the Commitment Shares. |
(2) |
The denominator is based on 20,985,784 shares outstanding as of June 30, 2023. The numerator is based on the number of shares issuable under the Purchase Agreement (that are the subject of this offering) at the corresponding assumed average purchase price set forth in the first column, which excludes the issuance of the Commitment Shares. |
(3) |
This number of shares reflects the Exchange Cap. We may only issue shares of our common stock in excess of the Exchange Cap if we obtain stockholder approval to do so, or if the average price of all shares of common stock issued to Lincoln Park under the Purchase Agreement equals or exceeds $4.399 per share. |
(4) |
The closing sale price of our common stock on The Nasdaq Capital Market on August 10, 2023. |
S-18
Pursuant to this prospectus supplement and the accompanying prospectus, we are offering up to $50,000,000 in shares of our common stock and 246,792 shares of our common stock are being issued to Lincoln Park as Commitment Shares pursuant to the Purchase Agreement. This prospectus supplement and the accompanying prospectus also cover the resale of these shares by Lincoln Park to the public.
We may, from time to time, and at our sole discretion, direct Lincoln Park to purchase shares of our common stock in a Regular Purchase in amounts up to the greater of 50,000 shares on any single business day from and after the Commencement provided that the closing sale price of our common stock is not below $0.25, which amounts may be increased to up to 60,000 shares or 70,000 shares of our common stock depending on the market price of our common stock at the time of sale, subject to, upon the parties mutual agreement, which share amounts and related market prices will be adjusted for any reorganization, recapitalization, non-cash dividend, stock split, reverse stock split or other similar transaction occurring after the date of the Purchase Agreement. In addition, upon notice to Lincoln Park, we may, from time to time and at our sole discretion, direct Lincoln Park to purchase additional shares of our common stock in “accelerated purchases,” and/or “additional accelerated purchases” as set forth in the Purchase Agreement. The purchase price per share is computed in accordance with the terms as set forth in the Purchase Agreement. Lincoln Park may not assign or transfer its rights and obligations under the Purchase Agreement. See “Lincoln Park Transaction — Purchases of Shares under the Purchase Agreement.”
Lincoln Park is an “underwriter” within the meaning of Section 2(a)(11) of the Securities Act. Lincoln Park has informed us that it will use an unaffiliated broker-dealer to effectuate all sales, if any, of the common stock that it may purchase from us pursuant to the Purchase Agreement. Such sales will be made on Nasdaq at prices and at terms then prevailing or at prices related to the then current market price. Each such unaffiliated broker-dealer will be an underwriter within the meaning of Section 2(a)(11) of the Securities Act. Lincoln Park has informed us that each such broker-dealer will receive commissions from Lincoln Park that will not exceed customary brokerage commissions.
We know of no existing arrangements between Lincoln Park and any other stockholder, broker, dealer, underwriter, or agent relating to the sale or distribution of the shares offered by this prospectus supplement.
We have paid $40,000 to Lincoln Park as Lincoln Park’s full and complete expense reimbursement in connection with entering into the transaction, including any legal fees.
We have agreed to indemnify Lincoln Park and certain other persons against certain liabilities in connection with the offering of shares of common stock offered hereby, including liabilities arising under the Securities Act or, if such indemnity is unavailable, to contribute amounts required to be paid in respect of such liabilities.
Lincoln Park represented to us that at no time prior to the date of the Purchase Agreement has Lincoln Park or its agents, representatives or affiliates engaged in or effected, in any manner whatsoever, directly or indirectly, any short sale (as such term is defined in Rule 200 of Regulation SHO of the Exchange Act) of our common stock or any hedging transaction, which establishes a net short position with respect to the common stock. Lincoln Park agreed that during the term of the Purchase Agreement, it, its agents, representatives or affiliates will not enter into or effect, directly or indirectly, any of the foregoing transactions.
We have advised Lincoln Park that it is required to comply with Regulation M promulgated under the Exchange Act. With certain exceptions, Regulation M precludes Lincoln Park, any affiliated purchasers, and any broker-dealer or other person who participates in the distribution from bidding for or purchasing, or attempting to induce any person to bid for or purchase any security which is the subject of the distribution until the entire distribution is complete. Regulation M also prohibits any bids or purchases made in order to stabilize the price of a security in connection with the distribution of that security. All of the foregoing may affect the marketability of the shares offered by this prospectus supplement.
This offering will terminate on the date that all shares offered by this prospectus supplement have been sold to Lincoln Park.
Our common stock is listed on The Nasdaq Capital Market and trades under the symbol “OCUP.” The transfer agent of our common stock is American Stock Transfer & Trust Company.
S-19
The validity of the issuance of our common stock offered in this prospectus will be passed upon for us by Honigman LLP, Kalamazoo, Michigan. Reed Smith LLP, New York, New York, is counsel for Lincoln Park in connection with this offering.
Ernst & Young LLP, independent registered public accounting firm, has audited our consolidated financial statements included in our Annual Report on Form 10-K for the year ended December 31, 2022, as set forth in their report, which is incorporated by reference in this prospectus and elsewhere in this registration statement. Our financial statements are incorporated by reference in reliance on Ernst & Young LLP’s report, given on their authority as experts in accounting and auditing.
This prospectus supplement and the accompanying prospectus form a part of a registration statement on Form S-3 that we filed with the SEC. This prospectus supplement and the accompanying prospectus do not contain all of the information set forth in the registration statement and the exhibits to the registration statement. For further information with respect to us and the securities we are offering under this prospectus supplement and the accompanying prospectus, we refer you to the registration statement and the exhibits and schedules filed as a part of the registration statement. You should rely only on the information contained in this prospectus supplement and the accompanying prospectus or incorporated by reference. We have not authorized anyone else to provide you with different information. We are not making an offer of these securities in any state where the offer is not permitted. You should not assume that the information in this prospectus supplement and the accompanying prospectus is accurate as of any date other than the date on the front page of this prospectus supplement, regardless of the time of delivery of this prospectus supplement and the accompanying prospectus or any sale of the securities offered by this prospectus supplement and the accompanying prospectus.
We are currently subject to the reporting requirements of the Exchange Act, and in accordance therewith, file periodic reports, proxy statements and other information with the SEC. The SEC maintains a website that contains reports, proxy and information statements and other information regarding issuers that file electronically with the SEC, including Ocuphire. The address of the SEC website is www.sec.gov. Our SEC filings are available to you on the SEC’s website at https://www.sec.gov and in the “Investor Relations” section of our website at https://ocuphire.com. Our website and the information contained on that site, or connected to that site, are not incorporated into and are not a part of this prospectus supplement and the accompanying prospectus.
S-20
The SEC allows us to “incorporate by reference” the information we file with it, which means that we can disclose important information to you by referring you to those documents instead of having to repeat the information in this prospectus. The information incorporated by reference is considered to be part of this prospectus, and later information that we file with the SEC will automatically update and supersede this information. We incorporate by reference the documents listed below and any future filings (including those made after the date of the initial filing of the registration statement of which this prospectus is a part and prior to the effectiveness of such registration statement) we will make with the SEC under Sections 13(a), 13(c), 14, or 15(d) of the Exchange Act until the termination of the offering of the shares covered by this prospectus (other than information furnished under Item 2.02 or Item 7.01 of Form 8-K):
• |
our Annual Report on Form 10-K for the year ended December 31, 2022, filed with the SEC on March 30, 2023, and the information specifically incorporated by reference into our Definitive Proxy Statement on Schedule 14A, filed with the SEC on May 1, 2023; |
• |
our Quarterly Reports on Form 10-Q for the quarters ended March 31, 2023 and June 30, 2023, filed with the SEC on May 15, 2023 and August 11, 2023, respectively; |
• |
our Current Reports on Form 8-K filed with the SEC on January 25, 2023, April 21, 2023, June 2, 2023, June 9, 2023, June 14, 2023, and August 11, 2023; and |
• |
the description of common stock set forth in the Registration Statement on Form 8-A, filed with the SEC on June 7, 2019, including any amendments thereto or reports filed for the purposes of updating this description, as updated by Exhibit 4.6 to our Annual Report on Form 10-K for the year ended December 31, 2022, filed with the SEC on March 30, 2023, including any amendments or reports filed for the purposes of updating this description. |
We will furnish without charge to each person, including any beneficial owner, to whom this prospectus supplement is delivered, upon written or oral request, a copy of any or all of the documents incorporated by reference, including exhibits to these documents. You may request a copy of these documents by writing or telephoning us at the following address:
Ocuphire Pharma, Inc.
37000 Grand River Avenue, Suite 120
Farmington Hills, MI 48335
(248) 957-9024
Attn: Rick Rodgers, Interim President and Chief Executive Officer
S-21
PROSPECTUS

$125,000,000
Common Stock
Preferred Stock
Debt Securities
Warrants
and
6,987,400 Shares of
Common Stock
Offered by the selling stockholders
We may, from time to time, offer and sell up to $125,000,000 of any combination of the securities described in this prospectus, either individually or in combination, at prices and on terms described in one or more supplements to this prospectus. We may also offer common stock or preferred stock upon conversion of debt securities, or common stock upon conversion of preferred stock, or common stock, preferred stock or debt securities upon exercise of warrants. In addition, the selling stockholders may offer and sell up to an aggregate of 6,987,400 shares of our common stock, from time to time, on the terms described in this prospectus or in an applicable prospectus supplement. We will not receive any of the proceeds from the sale of the shares offered by the selling stockholders hereunder. To the extent that any selling stockholder resells any securities, the selling stockholder may be required to provide you with this prospectus and a prospectus supplement identifying and containing specific information about the selling stockholders and the terms of the securities being offered.
This prospectus describes some of the general terms that may apply to an offering of our securities. We will provide the specific terms of these offerings and securities in one or more supplements to this prospectus. We may also authorize one or more free writing prospectuses to be provided to you in connection with these offerings. The prospectus supplement and any related free writing prospectus may also add, update or change information contained in this prospectus. You should carefully read this prospectus, the applicable prospectus supplement and any related free writing prospectus, as well as the documents incorporated by reference, before buying any of the securities being offered.
This prospectus may not be used to consummate a sale of securities unless accompanied by a prospectus supplement.
We and the selling stockholders may sell the securities through underwriters or dealers, directly to purchasers or through agents designated from time to time. For additional information on the methods of sale, you should refer to the section entitled “Plan of Distribution” in this prospectus on page 66 and in the applicable prospectus supplement. If any underwriters are involved in the sale of any securities with respect to which this prospectus is being delivered, the names of such underwriters and any applicable discounts or commissions and over-allotment options will be set forth in a prospectus supplement. The price to the public of such securities and the net proceeds we expect to receive from such sale will also be set forth in a prospectus supplement.
Our common stock is listed on the Nasdaq Capital Market under the symbol “OCUP.” On February 3, 2021, the last reported sale price of our common stock on the Nasdaq Capital Market was $13.31 per share. The applicable prospectus supplement will contain information, where applicable, as to other listings, if any, on the Nasdaq Capital Market or other securities exchange of the securities covered by the prospectus supplement.
Investing in our securities involves a high degree of risk. You should review carefully the risks and uncertainties described under the heading “Risk Factors” contained in this prospectus on page 9, in our most recent Annual Report on Form 10-K and Quarterly Report on Form 10-Q incorporated by reference into this prospectus, in the applicable prospectus supplement and in any free writing prospectuses we have authorized for use in connection with a specific offering, and under similar headings in the other documents that are incorporated by reference into this prospectus.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is February 12, 2021.
TABLE OF CONTENTS
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | | ||
| | |
You should rely only on the information contained in, or incorporated by reference into, this prospectus and the applicable prospectus supplement, along with the information contained in any free writing prospectuses we have authorized for use in connection with a specific offering. We have not authorized anyone to provide you with different information. We are not making an offer to sell or seeking an offer to buy securities under this prospectus or the applicable prospectus supplement and any related free writing prospectus in any jurisdiction where the offer or sale is not permitted. The information contained in this prospectus, the applicable prospectus supplement or any related free writing prospectus, and the documents incorporated by reference herein and therein, are accurate only as of their respective dates, regardless of the time of delivery of this prospectus, the applicable prospectus supplement or any related free writing prospectus, or any sale of a security.
i
This prospectus is part of a registration statement on Form S-3 that we filed with the Securities and Exchange Commission, or SEC, using a “shelf” registration process under the Securities Act of 1933, as amended, or the Securities Act. Under this shelf registration statement, we may sell from time to time in one or more offerings up to a total dollar amount of $125,000,000 of common stock and preferred stock, various series of debt securities and/or warrants to purchase any of such securities, either individually or in combination with other securities as described in this prospectus. In addition, under this shelf process, the selling stockholders to be named in a supplement to this prospectus may, from time to time, offer and sell up to 6,987,400 shares of our common stock, as described in this prospectus, in one or more offerings. This prospectus provides you with a general description of the securities we and the selling stockholders may offer.
Each time we or the selling stockholders sell any type or series of securities under this prospectus, we will provide a prospectus supplement that will contain more specific information about the terms of that offering. We may also authorize one or more free writing prospectuses to be provided to you that may contain material information relating to these offerings. We may also add, update or change in a prospectus supplement or free writing prospectus any of the information contained in this prospectus or in the documents we have incorporated by reference into this prospectus. This prospectus, together with the applicable prospectus supplement, any related free writing prospectus and the documents incorporated by reference into this prospectus and the applicable prospectus supplement, will include all material information relating to the applicable offering. You should carefully read both this prospectus and the applicable prospectus supplement and any related free writing prospectus, together with the additional information described under “Where You Can Find More Information,” before buying any of the securities being offered.
This prospectus may not be used to consummate a sale of securities unless accompanied by a prospectus supplement.
Neither we nor the selling stockholders have authorized anyone to provide you with any information other than contained in, or incorporated by reference into, this prospectus and the applicable prospectus supplement, along with the information contained in any free writing prospectuses we or the selling stockholders have authorized for use in connection with a specific offering. We take no responsibility for, and can provide no assurance as to the reliability of, any other information that others may give you. This prospectus is an offer to sell only the securities offered hereby, but only under circumstances and in jurisdictions where it is lawful to do so. You should not assume that the information contained in or incorporated by reference in this prospectus or any prospectus supplement or in any such free writing prospectus is accurate as of any date other than their respective dates. Our business, financial condition, results of operations and prospects may have changed since those dates.
This prospectus contains and incorporates by reference market data and industry statistics and forecasts that are based on independent industry publications and other publicly available information. Although we believe that these sources are reliable, we do not guarantee the accuracy or completeness of this information and we have not independently verified this information. Although we are not aware of any misstatements regarding the market and industry data presented in this prospectus and the documents incorporated herein by reference, these estimates involve risks and uncertainties and are subject to change based on various factors, including those discussed under the heading “Risk Factors” contained in the applicable prospectus supplement and any related free writing prospectus, and under similar headings in the other documents that are incorporated by reference into this prospectus. Accordingly, investors should not place undue reliance on this information.
This prospectus contains summaries of certain provisions contained in some of the documents described herein, but reference is made to the actual documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies of some of the documents referred to herein have been filed, will be filed or will be incorporated by reference as exhibits to the registration statement of which this prospectus is a part, and you may obtain copies of those documents as described below under the section entitled “Where You Can Find More Information.”
Except as otherwise indicated herein or as the context otherwise requires, references in this prospectus to “Ocuphire,” “the company,” “we,” “us,” “our” and similar references refer to Ocuphire Pharma, Inc., a corporation under the laws of the State of Delaware.
This prospectus and the information incorporated herein by reference include trademarks, service marks and trade names owned by us or other companies. All trademarks, service marks and trade names included or incorporated by reference into this prospectus, any applicable prospectus supplement or any related free writing prospectus are the property of their respective owners.
ii
This summary highlights selected information contained elsewhere in this prospectus or incorporated by reference herein and does not contain all of the information that you need to consider in making your investment decision. You should carefully read the entire prospectus, the applicable prospectus supplement and any related free writing prospectus, including the risks of investing in our securities discussed under the heading “Risk Factors” contained in this prospectus, the applicable prospectus supplement and any related free writing prospectus, and under similar headings in the other documents that are incorporated by reference into this prospectus. You should also carefully read the information incorporated by reference into this prospectus, including our financial statements and related notes, and the exhibits to the registration statement of which this prospectus is a part, before making your investment decision.
OCUPHIRE PHARMA, INC.
Overview
Ocuphire is a clinical-stage ophthalmic biopharmaceutical company focused on developing and commercializing therapies for the treatment of several eye disorders. Ocuphire’s pipeline currently includes two small molecule product candidates targeting front and back of the eye indications.
Its lead product candidate, Nyxol® Eye Drops (“Nyxol”), is a once-daily eye drop formulation of phentolamine mesylate designed to reduce pupil diameter and improve visual acuity. As a result, Nyxol can potentially be used for the treatment of multiple indications such as dim light or night vision disturbances (“NVD”), pharmacologically-induced mydriasis (which refers to the use of pharmacological agents to dilate the pupil for office-based eye exams) and presbyopia (a gradual, age-related loss of the eyes’ ability to focus on nearby objects). Ocuphire management believes this multiple indication potential represents a significant market opportunity. Nyxol has been studied across three Phase 1 and four Phase 2 trials totaling over 230 patients and has demonstrated promising clinical data for use in multiple ophthalmic indications. Ocuphire initiated a Phase 3 trial for the treatment of NVD in the fourth quarter of 2020, a Phase 3 trial for reversal of pharmacologically-induced mydriasis (“RM”) in the fourth quarter of 2020, and plans to initiate a Phase 2 trial in combination with low dose pilocarpine for presbyopia, in the first quarter of 2021. Ocuphire expects top-line results to read out as early as the first quarter of 2021 and throughout the remainder of 2021, and, assuming successful and timely completion of further trials, anticipates submitting a new drug application (“NDA”) to the U.S. Food and Drug Administration (“FDA”) in early 2023 under the 505(b)(2) pathway.
Ocuphire’s second product candidate, APX3330, is a twice-a-day oral tablet, designed to target multiple pathways relevant to retinal and choroidal (the vascular layer of the eye) vascular diseases, such as diabetic retinopathy (“DR”) and diabetic macular edema (“DME”) which if left untreated may result in permanent visual acuity loss and eventual blindness. DR is a disease resulting from diabetes, in which chronically elevated blood sugar levels cause progressive damage to blood vessels in the retina. DME is a severe form of DR which involves leakage of protein and fluid into the macula, the central portion of the retina, causing swelling. Prior to Ocuphire’s in-licensing of the product candidate, APX3330 had been studied by third parties in six Phase 1 and five Phase 2 trials totaling over 440 patients, for inflammatory and oncology indications, and had demonstrated promising evidence of tolerability, pharmacokinetics, durability and target engagement. Ocuphire plans to initiate a Phase 2 trial for APX3330 in the first quarter of 2021 for the treatment of patients with DR, including moderately severe non-proliferative DR (“NPDR”) and mild proliferative DR (“PDR”), as well as patients with DME without loss of central vision. Ocuphire has also in-licensed additional second generation product candidates, analogs of APX3330, including APX2009 and APX2014.
As part of its strategy, Ocuphire will continue to explore opportunities to acquire additional ophthalmic assets and to seek strategic partners for late stage development, regulatory preparation and commercialization of drugs in key global markets.
Ocuphire estimates that there are 15-20 million moderate-to-severe NVD patients in the United States, over 80 million eye exams conducted per year with pharmacologically-induced mydriasis, over 100 million presbyopia patients, over 7 million patients with DR, and 750,000 patients with DME. There are no currently approved pharmacological products on the market for NVD, RM, or presbyopia. In the case of presbyopia there are non-pharmacologic and potentially inconvenient treatments such as reading glasses or contact lenses, as well as invasive surgical interventions with associated risks such as creation or worsening of NVD. For DR and DME, intraocular injections targeting vascular endothelial growth factors (“VEGF”) (a family of proteins that promote
1
angiogenesis – the formation of new blood vessels – and vascular permeability) are approved globally, but these chronic therapies require frequent biweekly or monthly office visits and are prone to side effects such as hemorrhage, intraocular infection, and increased risk of blood clots.
Ocuphire is developing Nyxol and APX3330 for multiple indications. Ocuphire believes the two programs present similar potential advantages: (1) promising clinical data to date; (2) small molecules; (3) convenient dosing route and schedule; (4) potential for first-line or adjunct therapy; and (5) significant commercial potential. In the fourth quarter of 2020, Ocuphire initiated Phase 3 clinical trials for Nyxol in NVD and RM, with announcement of completion of enrollment in Phase 3 RM trial in the fourth quarter of 2020. In the first quarter of 2021. Ocuphire expects to initiate a Phase 2 proof of concept trial in presbyopia for a kit combination of Nyxol and low-dose pilocarpine, a pupil constrictor with a mechanism different and complementary to Nyxol. In preparation for at least one of the two Phase 3 registration trials for Nyxol, Ocuphire plans to launch a blow-fill-seal manufacturing program for preservative-free single use Nyxol eye drops. Furthermore, Ocuphire plans to initiate a 6-month rabbit toxicology study in the first quarter of 2021, completion of which is necessary prior to commencement of the Phase 3 safety exposure trial for chronic indications. Ocuphire also expects to launch a Phase 2 trial for APX3330 in DR and DME in the first quarter of 2021 with a concurrent Phase 2/3 oral tablet manufacturing program. TABLE 1 below summarizes Ocuphire’s current development pipeline of product candidates and their target indications:
TABLE 1. Ocuphire Pipeline Indications
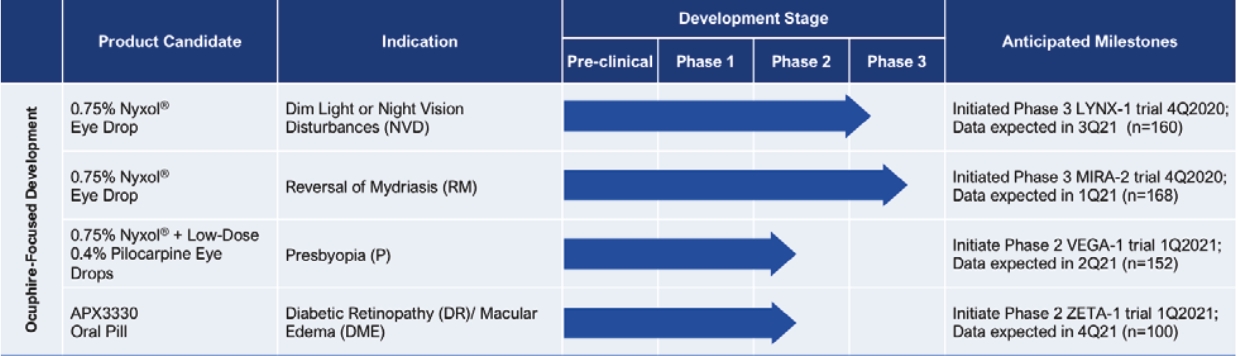
Note: 0.75% Nyxol (Phentolamine Ophthalmic Solution) is the same as 1% Nyxol (Phentolamine Mesylate Ophthalmic Solution).
The Merger, Reverse Stock Split and Name Change
On November 5, 2020, Ocuphire (formerly known as Rexahn Pharmaceuticals, Inc., and prior to the merger, referred to as “Rexahn”), completed its business combination with Ocuphire Pharma, Inc. (“Ocuphire Private Company”), in accordance with the terms of the Agreement and Plan of Merger, dated as of June 17, 2020, as amended, by and among Rexahn, Ocuphire Private Company, and Razor Merger Sub, Inc., a wholly-owned subsidiary of Rexahn (“Merger Sub”) (as amended, the “Merger Agreement”), pursuant to which Merger Sub merged with and into Ocuphire Private Company, with Ocuphire Private Company surviving as a wholly owned subsidiary of Rexahn (the “Merger”).
In connection with, and immediately prior to the completion of, the Merger, Rexahn effected a reverse stock split of the common stock, at a ratio of 1-for-4 (the “Reverse Stock Split”). Under the terms of the Merger Agreement, after taking into account the Reverse Stock Split, Rexahn issued shares of its common stock to Ocuphire Private Company stockholders, based on a common stock exchange ratio of 1.0565 shares of common stock for each share of Ocuphire Private Company common stock. In connection with the Merger, Rexahn changed its name from “Rexahn Pharmaceuticals, Inc.” to “Ocuphire Pharma, Inc.,” and the business conducted by Rexahn became the business conducted by Ocuphire Private Company.
Private Placement of Common Shares and Warrants
On November 5, 2020, Ocuphire and Ocuphire Sub completed a private placement transaction (the “Pre-Merger Financing”) with certain accredited investors (the “Investors”) pursuant to that certain Amended and Restated Securities Purchase Agreement (the “Securities Purchase Agreement”) dated June 29, 2020 by and among the Company, Ocuphire Sub and the Investors for an aggregate purchase price of approximately $21.15 million (the “Purchase Price”).
2
Pursuant to the Pre-Merger Financing, (i) Ocuphire Sub issued and sold to the Investors shares of Ocuphire Sub’s common stock (the “Initial Shares”) which converted pursuant to the exchange ratio in the Merger into an aggregate of approximately 1,249,996 shares (the “Converted Initial Shares”) of the Company’s common stock, (ii) Ocuphire Sub deposited into escrow, for the benefit of the Investors, additional shares of Ocuphire Sub’s common stock (the “Additional Shares”) which converted pursuant to the exchange ratio in the Merger into an aggregate of approximately 3,749,992 shares of common stock (the “Converted Additional Shares”), which Converted Additional Shares were delivered (or became deliverable) to the Investors on November 19, 2020, and (iii) the Company agreed to issue to each Investor on the tenth trading day following the consummation of the Merger Series A Warrants representing the right to acquire shares of common stock equal to the sum of (a) the Converted Initial Shares purchased by the Investor, (b) the Converted Additional Shares delivered or deliverable to the Investor, without giving effect to any limitation on delivery contained in the Securities Purchase Agreement and (c) the number of shares of Common Stock, if any, underlying the Series B Warrants issued to the Investor (the “Series A Warrants”) and additional Series B Warrants to purchase shares of Common Stock (the “Series B Warrants” and together with the Series A Warrants, the “Series A/B Warrants”).
On November 19, 2020 (the “Warrant Closing Date”), pursuant to the terms of the Amended and Restated Securities Purchase Agreement, the Company issued the Series A Warrants and the Series B Warrants. For a description of the Series A Warrants and Series B Warrants, see “Description of Capital Stock—Outstanding Warrants.”
On February 3, 2021, the Company entered into a Waiver Agreement with each of the investors (collectively, the “Waiver Agreements”). Pursuant to the Waiver Agreements, the investors and the Company agreed to waive certain rights, finalize the exercise price and number of Warrants, and, in the case of certain major holders (“Major Holders”), grant certain registration rights for the shares underlying the Warrants as described herein.
Waiver of Warrant Resets and Final Determination of Number of Series B Warrants
Pursuant to the Waiver Agreements, each investor agreed to waive the reset provisions in the Warrants in order to allow for one more immediate and final reset of the number of shares of common stock underlying the Warrants and the exercise price of the Series A Warrants such that (A) the exercise price of the Series A Warrants was fixed at the initial exercise price of $4.4795 per share, (B) the number of shares underlying all of the Series A Warrants was fixed at the initial number of 5,665,838 in the aggregate and (C) in the case of all Holders, the number of shares underlying all of the Series B Warrants was fixed at 1,708,334 in the aggregate with respect to such Holders (the “Final Series B Reset”).
The directors and the officers waived their right to the Final Series B Reset such that the number of Series B Warrants for such directors and officers was fixed at the initial number of Series B Warrants issued to them on November 19, 2020.
The Waiver Agreement provides that the Company may file this registration statement on Form S-3, and may further file registration statements and undertake Subsequent Placements at any time following the close of business on March 1, 2021, subject to the Major Holders’ participation rights.
In connection with the Waiver Agreement, each of the Major Holder’s participation rights were extended to December 31, 2021, subject to a limitation of participation in any such Subsequent Placement (50% in the aggregate); provided that such participation rights will not apply to any at-the-market equity offering implemented by the Company through a broker dealer at any time following the Effective Date; and provided further that the Company will not be able to issue any equity securities under such at-the-market equity offering until on or after April 15, 2021.
Risks Associated with our Business
Our business is subject to numerous risks, as described under the heading “Risk Factors” contained in the applicable prospectus supplement and in any free writing prospectuses we have authorized for use in connection with a specific offering, and under similar headings in the documents that are incorporated by reference into this prospectus.
Description of Securities We May Offer
We may offer shares of our common stock and preferred stock, various series of debt securities and/or warrants to purchase any of such securities, either individually or in combination, with a total dollar amount up to $125,000,000 from time to time under this prospectus, together with the applicable prospectus supplement and any related free writing prospectus, at prices and on terms to be determined at the time of any offering. We may also offer common
3
stock, preferred stock and/or debt securities upon the exercise of warrants. In addition, the selling stockholders to be named in a supplement to this prospectus may offer or sell, from time to time, up to 6,987,400 shares of our common stock. This prospectus provides you with a general description of the securities we may offer. Each time we or the selling stockholders offer a type or series of securities under this prospectus, we will provide a prospectus supplement that will describe the specific amounts, prices and other important terms of the securities, including, to the extent applicable:
• |
designation or classification; |
• |
aggregate principal amount or aggregate offering price; |
• |
maturity; |
• |
original issue discount; |
• |
rates and times of payment of interest or dividends; |
• |
redemption, conversion, exercise, exchange or sinking fund terms; |
• |
ranking; |
• |
restrictive covenants; |
• |
voting or other rights; |
• |
conversion or exchange prices or rates and, if applicable, any provisions for changes to or adjustments in the conversion or exchange prices or rates and in the securities or other property receivable upon conversion or exchange; and |
• |
a discussion of material United States federal income tax considerations, if any. |
The applicable prospectus supplement and any related free writing prospectus that we may authorize to be provided to you may also add, update or change any of the information contained in this prospectus or in the documents we have incorporated by reference. However, no prospectus supplement or free writing prospectus will offer a security that is not registered and described in this prospectus at the time of the effectiveness of the registration statement of which this prospectus is a part.
This prospectus may not be used to consummate a sale of securities unless it is accompanied by a prospectus supplement.
We or the selling stockholders may sell the securities directly to investors or to or through agents, underwriters or dealers. We and the selling stockholders, and our or their agents, underwriters or dealers reserve the right to accept or reject all or part of any proposed purchase of securities. If we do offer securities to or through agents, underwriters or dealers, we will include in the applicable prospectus supplement:
• |
the names of those agents, underwriters or dealers; |
• |
applicable fees, discounts and commissions to be paid to them; |
• |
details regarding over-allotment options, if any; and |
• |
the net proceeds to us, if any. |
Common Stock
We may issue shares of our common stock from time to time. The selling stockholders may offer shares of our common stock to the extent such shares were issued and outstanding, or issuable upon exercise or conversion of securities issued and outstanding, prior to the original date of filing of the registration statement to which this prospectus relates. Each holder of our common stock is entitled to one vote for each share on all matters submitted to a vote of the stockholders, including the election of directors. Under our amended and restated certificate of incorporation, as amended, or certificate of incorporation, and second amended and restated bylaws, or bylaws, our stockholders do not have cumulative voting rights. Because of this, the holders of a majority of the shares of common stock entitled to vote in any election of directors can elect all of the directors standing for election, if they should so choose. Subject to preferences that may be applicable to any then-outstanding preferred stock, holders of common stock are entitled to receive ratably those dividends, if any, as may be declared from time to time by our board of
4
directors out of legally available funds. In the event of our liquidation, dissolution or winding up, holders of common stock are entitled to share ratably in the net assets legally available for distribution to stockholders after the payment of all of our debts and other liabilities and the satisfaction of any liquidation preference granted to the holders of any then-outstanding shares of preferred stock. Holders of common stock have no preemptive, conversion or subscription rights and there are no redemption or sinking fund provisions applicable to the common stock. The rights, preferences and privileges of the holders of common stock are subject to, and may be adversely affected by, the rights of the holders of shares of any series of preferred stock that we may designate in the future. In this prospectus, we have summarized certain general features of the common stock under “Description of Capital Stock—Common Stock.” We urge you, however, to read the applicable prospectus supplement (and any related free writing prospectus that we may authorize to be provided to you) related to any common stock being offered.
Preferred Stock
We may issue shares of our preferred stock from time to time, in one or more series. Our board of directors will determine the designations, voting powers, preferences and rights of the preferred stock, as well as the qualifications, limitations or restrictions thereof, including dividend rights, conversion rights, preemptive rights, terms of redemption or repurchase, liquidation preferences, sinking fund terms and the number of shares constituting any series or the designation of any series, or the designation of such series, any or all of which may be greater than the rights of our common stock. Convertible preferred stock will be convertible into our common stock or exchangeable for other securities. Conversion may be mandatory or at your option and would be at prescribed conversion rates.
If we sell any series of preferred stock under this prospectus, we will fix the designations, voting powers, preferences and rights of such series of preferred stock, as well as the qualifications, limitations or restrictions thereof, in the certificate of designation relating to that series. We will file as an exhibit to the registration statement of which this prospectus is a part, or will incorporate by reference from reports that we file with the SEC, the form of any certificate of designation that describes the terms of the series of preferred stock that we are offering before the issuance of the related series of preferred stock. In this prospectus, we have summarized certain general features of the preferred stock under “Description of Capital Stock—Preferred Stock.” We urge you to read the applicable prospectus supplement (and any free writing prospectus that we may authorize to be provided to you) related to the series of preferred stock being offered, as well as the complete certificate of designation that contains the terms of the applicable series of preferred stock.
Debt Securities
We may issue debt securities from time to time, in one or more series, as either senior or subordinated debt or as senior or subordinated convertible debt. The senior debt securities will rank equally with any other unsecured and unsubordinated debt. The subordinated debt securities will be subordinate and junior in right of payment, to the extent and in the manner described in the instrument governing the debt, to all of our senior indebtedness. Convertible or exchangeable debt securities will be convertible into or exchangeable for our common stock or our other securities. Conversion or exchange may be mandatory or optional (at our option or the holders’ option) and would be at prescribed conversion or exchange rates.
Any debt securities issued under this prospectus will be issued under one or more documents called indentures, which are contracts between us and a national banking association or other eligible party, as trustee. In this prospectus, we have summarized certain general features of the debt securities under “Description of Debt Securities.” We urge you, however, to read the applicable prospectus supplement (and any free writing prospectus that we may authorize to be provided to you) related to the series of debt securities being offered, as well as the complete indenture and any supplemental indentures that contain the terms of the debt securities. A form of indenture has been filed as an exhibit to the registration statement of which this prospectus is a part, and supplemental indentures and forms of debt securities containing the terms of the debt securities being offered will be filed as exhibits to the registration statement of which this prospectus is a part or will be incorporated by reference from reports that we file with the SEC.
Warrants
We may issue warrants for the purchase of common stock, preferred stock and/or debt securities in one or more series. We may issue warrants independently or in combination with common stock, preferred stock and/or debt securities offered by any prospectus supplement. In this prospectus, we have summarized certain general features of the warrants under “Description of Warrants.” We urge you, however, to read the applicable prospectus supplement
5
(and any related free writing prospectus that we may authorize to be provided to you) related to the particular series of warrants being offered, as well as any warrant agreements and warrant certificates, as applicable, that contain the terms of the warrants. We have filed forms of the warrant agreements and forms of warrant certificates containing the terms of the warrants that may be offered as exhibits to the registration statement of which this prospectus is a part. We will file as exhibits to the registration statement of which this prospectus is a part, or will incorporate by reference from reports that we file with the SEC, the form of warrant and/or the warrant agreement and warrant certificate, as applicable, that contain the terms of the particular series of warrants we are offering, and any supplemental agreements, before the issuance of such warrants.
Any warrants issued under this prospectus may be evidenced by warrant certificates. Warrants also may be issued under an applicable warrant agreement that we enter into with a warrant agent. We will indicate the name and address of the warrant agent, if applicable, in the prospectus supplement relating to the particular series of warrants being offered.
Use of Proceeds
Except as described in any applicable prospectus supplement or in any free writing prospectuses we have authorized for use in connection with a specific offering, we currently intend to use the net proceeds from this offering for working capital and general corporate purposes, which may include, among other things, funding research and development programs, vendor payables, hiring additional personnel, and capital expenditures.
We will not receive any proceeds from the sale of shares of our common stock by any selling stockholders.
Selling Stockholders
The selling stockholders acquired Series A Warrants and Series B Warrants exercisable for shares of our common stock in November 2020. See the section entitled “Selling Stockholders” on page 64 of this prospectus.
Company Information
Our principal executive offices are located at 37000 Grand River Avenue, Suite 120, Farmington Hills, MI 48335. Our telephone number is (248) 681-9815. Our website address is www.ocuphire.com. The information contained in, or accessible through, our website does not constitute part of this prospectus, should not be relied on in determining whether to make an investment decision, and the inclusion of our website address in this prospectus is an inactive textual reference only.
6
Investing in our securities involves a high degree of risk. The following is a summary of the principal risks and uncertainties that could materially adversely affect our business, financial condition, or results of operations. You should read this summary together with the more detailed description of risk factors contained under the heading “Risk Factors” in this prospectus on page 9, as well as in the applicable prospectus supplement and in any free writing prospectuses we have authorized for use in connection with a specific offering.
• |
Ocuphire currently depends entirely on the success of Nyxol and APX3330, its only product candidates. Ocuphire may never receive marketing approval for, or successfully commercialize, Nyxol, APX3330, or other product candidates it may pursue in the future for any indication. |
• |
The results of previous clinical trials may not be predictive of future results, and the results of Ocuphire’s current and planned clinical trials may not satisfy the requirements of the FDA or non-U.S. regulatory authorities. |
• |
Changes in regulatory requirements or FDA guidance, or unanticipated events during Ocuphire’s clinical trials, may result in changes to clinical trial protocols or additional clinical trial requirements, which could result in increased costs to Ocuphire or delays in its development timeline. |
• |
Ocuphire has incurred only losses since inception. Ocuphire expects to incur losses for the foreseeable future and may never achieve or maintain profitability. |
• |
Ocuphire’s recurring operating losses have raised substantial doubt regarding its ability to continue as a going concern. |
• |
Raising additional capital may cause dilution to Ocuphire’s stockholders, restrict Ocuphire’s operations, or require Ocuphire to relinquish rights to its technologies or product candidates. |
• |
Even if it receives marketing approval for its product candidates in the United States, Ocuphire may never receive regulatory approval to market such product candidates outside of the United States. |
• |
Even if Ocuphire obtains marketing approval for its product candidates, such product candidates could be subject to post-marketing restrictions or withdrawal from the market, and Ocuphire may be subject to substantial penalties if it fails to comply with regulatory requirements or experience unanticipated problems with a product following approval. |
• |
Ocuphire’s relationships with healthcare providers and third-party payors will be subject to applicable fraud and abuse and other healthcare laws and regulations, which could expose Ocuphire to criminal sanctions, civil penalties, contractual damages, reputational harm, and diminished profits and future earnings, among other penalties and consequences. |
• |
Ocuphire employees may engage in misconduct or other improper activities, including violating applicable regulatory standards and requirements or engaging in insider trading, which could significantly harm Ocuphire’s business. |
• |
Ocuphire faces substantial competition, which may result in others discovering, developing, or commercializing products before or more successfully than it does. |
• |
Ocuphire lacks experience in commercializing products, which may have an adverse effect on its business. |
• |
If Ocuphire is unable to establish sales and marketing capabilities or enter into agreements with third parties to sell, market, and distribute its product candidates, if approved, it may not be successful in commercializing such product candidates if and when they are approved. |
• |
Even if Ocuphire is able to commercialize its product candidates, their profitability will likely depend in significant part on third-party reimbursement practices, which, if unfavorable, would harm its business. |
• |
Product liability lawsuits against Ocuphire, or its suppliers and manufacturers, could cause it to incur substantial liabilities and could limit commercialization of any product candidate that it may develop. |
• |
Ocuphire will be unable to directly control all aspects of its clinical trials due to its reliance on clinical research organizations (CROs) and other third parties that assist Ocuphire in conducting clinical trials. |
7
• |
If Ocuphire is not able to establish new collaborations on commercially reasonable terms, it may have to alter its development, manufacturing, and commercialization plans. |
• |
If Ocuphire is unable to obtain and maintain sufficient patent protection for its product candidates, its competitors could develop and commercialize products or technology similar or identical to those of Ocuphire, which would adversely affect Ocuphire’s ability to successfully commercialize any product candidates it may develop, its business, results of operations, financial condition and prospects. |
• |
If Ocuphire does not obtain protection under the Hatch-Waxman Act and similar foreign legislation by extending the patent terms and obtaining data exclusivity for its product candidate, its business may be materially harmed. |
• |
Changes in U.S. patent law could diminish the value of patents in general, thereby impairing Ocuphire’s ability to protect its product candidates. |
• |
Ocuphire may not be able to protect or practice its intellectual property rights throughout the world. |
• |
Obtaining and maintaining Ocuphire’s patent protection depends on compliance with various procedural, document submission, fee payment, and other requirements imposed by governmental agencies, and its patent protection could be reduced or eliminated for noncompliance with these requirements. |
• |
Ocuphire depends on intellectual property sublicensed from Apexian Pharmaceuticals, Inc. (“Apexian”) for its APX3330 product candidate under development and its additional pipeline candidates, and the termination of, or reduction or loss of rights under, this sublicense would harm Ocuphire’s business. |
• |
Ocuphire is dependent on its key personnel, and if it is not successful in attracting and retaining highly qualified personnel, it may not be able to successfully implement its business strategy. |
• |
Ocuphire will need to develop and expand its company, and may encounter difficulties in managing this development and expansion, which could disrupt its operations. |
• |
The COVID-19 pandemic has and could continue to adversely impact Ocuphire’s business, including pre-clinical and clinical trials and regulatory approvals. |
• |
Ocuphire’s insurance policies are expensive and protect only from some business risk, which leaves Ocuphire exposed to significant uninsured liabilities. |
• |
Ocuphire does not anticipate paying any cash dividends in the foreseeable future. |
• |
If Ocuphire fails to comply with the continued listing standards of the Nasdaq Capital Market, Ocuphire common stock could be delisted. If it is delisted, Ocuphire common stock and the liquidity of its common stock would be impacted. |
• |
The market price of Ocuphire common stock may fluctuate significantly. |
• |
Ocuphire may be subject to securities litigation, which is expensive and could divert management attention. |
8
Investing in our securities involves a high degree of risk. Before deciding whether to invest in our securities, you should consider carefully the risks and uncertainties described below, under the heading “Risk Factors” contained in the applicable prospectus supplement and any related free writing prospectuses we have authorized for use in connection with a specific offering, and discussed under the section entitled “Risk Factors” contained in our most recent Annual Report on Form 10-K and in our most recent Quarterly Report on Form 10-Q, as well as any amendments thereto reflected in subsequent filings with the SEC, which are incorporated by reference into this prospectus in their entirety, together with other information in this prospectus, the documents incorporated by reference and any free writing prospectus that we may authorize for use in connection with this offering.
The risks described in these documents are not the only ones we face, but those that we consider to be material. There may be other unknown or unpredictable economic, business, competitive, regulatory or other factors that could have material adverse effects on our future results. Past financial performance may not be a reliable indicator of future performance, and historical trends should not be used to anticipate results or trends in future periods. If any of these risks actually occurs, our business, financial condition, results of operations or cash flow could be seriously harmed. This could cause the trading price of our common stock to decline, resulting in a loss of all or part of your investment. Please also read carefully the section below entitled “Forward-Looking Statements.”
Risks Related to Development of Ocuphire’s Product Candidates
Ocuphire currently depends entirely on the success of Nyxol and APX3330, its only product candidates. Ocuphire may never receive marketing approval for, or successfully commercialize, Nyxol, APX3330, or other product candidates it may pursue in the future for any indication.
Ocuphire currently has only two product candidates, Nyxol and APX3330, in clinical development, and its business depends on their successful clinical development, regulatory approval and commercialization. The research, testing, manufacturing, labeling, approval, sale, marketing and distribution of a drug product are subject to extensive regulation by the FDA and other regulatory authorities in the United States and other countries, where regulations may differ. Ocuphire is not permitted to market its product candidates in the United States until it receives approval of an NDA from the FDA or in any foreign countries until it receives the requisite approval from such countries. Ocuphire has not submitted an NDA to the FDA or comparable applications to other regulatory authorities or received marketing approval for its product candidates. Before obtaining regulatory approval for the commercial sale of its product candidates for a particular indication, Ocuphire must demonstrate through preclinical testing and clinical trials that the applicable product candidate is safe and effective for use in that target indication. This process can take many years and may be followed by post-marketing studies and surveillance together which will require the expenditure of substantial resources beyond the proceeds raised in the Pre-Merger Financing. Of the large number of drugs in development in the United States, only a small percentage of drugs successfully complete the FDA regulatory approval process and are commercialized. Accordingly, even if Ocuphire is able to complete development of its product candidates, Ocuphire cannot assure you that its product candidates will be approved or commercialized.
Obtaining approval of an NDA is an extensive, lengthy, expensive and uncertain process, and the FDA may delay, limit or deny approval of Ocuphire’s product candidates for many reasons, including:
• |
the data collected from preclinical studies and clinical trials of Ocuphire’s product candidates may not be sufficient to support the submission of an NDA; |
• |
Ocuphire may not be able to demonstrate to the satisfaction of the FDA that its product candidates are safe and effective for any indication; |
• |
the results of clinical trials may not meet the level of statistical significance or clinical significance required by the FDA for approval; |
• |
the FDA may disagree with the number, design, size, conduct, or implementation of Ocuphire’s clinical trials; |
• |
the FDA may not find the data from preclinical studies and clinical trials sufficient to demonstrate that Ocuphire’s product candidates’ clinical and other benefits outweigh the safety risks; |
• |
the FDA may disagree with Ocuphire’s interpretation of data from preclinical studies or clinical trials; |
• |
the FDA may not accept data generated at Ocuphire’s clinical trial sites; |
9
• |
the FDA may have difficulties scheduling an advisory committee meeting in a timely manner or the advisory committee may recommend against approval of Ocuphire’s application or may recommend that the FDA require, as a condition of approval, additional preclinical studies or clinical trials, limitations on approved labeling or distribution and use restrictions; |
• |
the FDA may require development of a Risk Evaluation and Mitigation Strategy (REMS) as a condition of approval; |
• |
the FDA may identify deficiencies in the manufacturing processes or facilities of third party manufacturers with which Ocuphire enters into agreements for clinical and commercial supplies; or |
• |
the FDA may change its approval policies or adopt new regulations. |
The results of previous clinical trials may not be predictive of future results, and the results of Ocuphire’s current and planned clinical trials may not satisfy the requirements of the FDA or non-U.S. regulatory authorities.
The results from the prior preclinical studies and clinical trials for Nyxol and APX3330 discussed elsewhere in this prospectus may not necessarily be predictive of the results of future preclinical studies or clinical trials. Even if Ocuphire is able to complete its planned clinical trials of its product candidates according to its current development timeline, the results from its prior clinical trials of its product candidates may not be replicated in these future trials. Many companies in the pharmaceutical and biotechnology industries (including those with greater resources and experience than Ocuphire) have suffered significant setbacks in late-stage clinical trials after achieving positive results in early stage development, and Ocuphire cannot be certain that it will not face similar setbacks. These setbacks have been caused by, among other things, preclinical findings made while clinical trials were underway or safety or efficacy observations made in clinical trials, including previously unreported adverse events (“AEs”). Moreover, preclinical and clinical data are often susceptible to varying interpretations and analyses, and many companies that believed their product candidates performed satisfactorily in preclinical studies and clinical trials nonetheless have failed to obtain FDA approval. Additionally, Ocuphire is developing, as a treatment for Presbyopia, a combination product candidate of Nyxol and low-dose pilocarpine in a two-part kit, which have not been studied together yet. If Ocuphire fails to produce positive results in its clinical trials of any of its product candidates, the development timelines and regulatory approvals and commercialization prospects for its product candidates and its business and financial prospects, would be adversely affected. If Ocuphire fails to produce positive results in its clinical trials of any of its product candidates, the development timelines, regulatory approvals, and commercialization prospects for its product candidates, as well as Ocuphire’s business and financial prospects, would be adversely affected. Further, Ocuphire’s product candidates may not be approved even if they achieve their respective primary endpoints in Phase 3 registration trials. The FDA or non-U.S. regulatory authorities may disagree with Ocuphire’s trial designs or its interpretation of data from preclinical studies and clinical trials. In addition, any of these regulatory authorities may change requirements for the approval of a product candidate even after reviewing and providing comments or advice on a protocol for a pivotal clinical trial that has the potential to result in approval by the FDA or another regulatory authority. Furthermore, any of these regulatory authorities may also approve Ocuphire’s product candidate for fewer or more limited indications than it requests or may grant approval contingent on the performance of costly post-marketing clinical trials.
Ocuphire completed two Phase 2b clinical trials for Nyxol in patients with pharmacologically induced mydriasis and in elderly patients with ocular hypertension (“OHT”) in the second half of 2019. For Nyxol, Ocuphire commenced a Phase 3 trial for the treatment of NVD in the fourth quarter of 2020, a Phase 3 trial for RM in the fourth quarter of 2020, and plans to commence a Phase 2 trial in combination with low-dose pilocarpine for presbyopia, in the first quarter of 2021. For APX3330, Ocuphire plans to commence a Phase 2 trial for the treatment of patients with DR, including patients with moderately severe NPDR and mild PDR, as well as patients with DME without loss of central vision, in the first quarter of 2021. Ocuphire also plans to pursue further clinical and preclinical trials as described elsewhere in this prospectus. If successful, Ocuphire plans to eventually seek regulatory approvals of Nyxol and APX3330 initially in the United States, Canada, and Europe, and may seek approvals in other geographies. Before obtaining regulatory approvals for the commercial sale of any product candidate for any target indication, Ocuphire must demonstrate with substantial evidence gathered in preclinical studies and adequate and well-controlled clinical studies, and, with respect to approval in the United States, to the satisfaction of the FDA, that the product candidate is safe and effective for use for that target indication. Ocuphire cannot assure you that the FDA or non-U.S. regulatory authorities would consider its planned clinical trials to be sufficient to serve as the basis for approval of its product candidates for any indication. The FDA and non-U.S. regulatory authorities retain broad discretion in evaluating the results of Ocuphire’s clinical trials and in determining whether the results demonstrate that its product
10
candidates are safe and effective. If Ocuphire is required to conduct clinical trials of its product candidates in addition to those it has planned prior to approval, Ocuphire will need substantial additional funds, and cannot assure you that the results of any such outcomes trial or other clinical trials will be sufficient for approval.
If clinical trials of Ocuphire’s product candidates fail to demonstrate safety and efficacy to the satisfaction of regulatory authorities or do not otherwise produce positive results, Ocuphire may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development and commercialization of such product candidates.
Before obtaining marketing approval from regulatory authorities for the sale of Nyxol, Ocuphire must complete additional Phase 2 and Phase 3 clinical trials to demonstrate the safety and efficacy in humans. Additionally, for chronic indication Ocuphire must complete a six-month toxicology study in rabbits. Clinical testing is expensive, difficult to design and implement, can take many years to complete, and is uncertain as to outcome. A failure of one or more clinical trials can occur at any stage of development. In addition, based on the Phase 2 safety, tolerability and efficacy results of APX3330 in patients with DR/DME, Ocuphire might need further animal toxicology studies and additional Phase 2 and Phase 3 clinical trials before obtaining marketing approval from regulatory authorities for the sale of APX3330.
Ocuphire, or its future collaborators, may experience numerous unforeseen events during, or as a result of, clinical trials that could result in increased development costs and delay, and could limit or prevent its ability to receive marketing approval or commercialize its product candidates, including:
• |
regulators or IRBs may not authorize Ocuphire or its investigators to commence a clinical trial or conduct a clinical trial at a prospective trial site including due to the ongoing COVID-19 pandemic or other public health emergency; |
• |
government or regulatory delays and changes in regulatory requirements, policy and guidelines may require Ocuphire to perform additional clinical trials or use substantial additional resources to obtain regulatory approval, including due to the ongoing COVID-19 pandemic or other public health emergency; |
• |
Ocuphire may have delays in reaching or fail to reach agreement on acceptable clinical trial contracts or clinical trial protocols with prospective trial sites, including due to the ongoing COVID-19 pandemic or other public health emergency; |
• |
clinical trials may produce negative or inconclusive results, and Ocuphire may decide, or regulators may require it, to conduct additional clinical trials or abandon product development programs, including due to the ongoing COVID-19 pandemic or other public health emergency; |
• |
the number of patients required for clinical trials may be larger, enrollment in these clinical trials may be slower or participants may drop out of these clinical trials at a higher rate than Ocuphire anticipates, including due to the ongoing COVID-19 pandemic or other public health emergency; |
• |
Ocuphire’s third-party contractors may fail to comply with regulatory requirements or meet their contractual obligations to Ocuphire in a timely manner, or at all; |
• |
Ocuphire’s patients or medical investigators may be unwilling to follow its clinical trial protocols; |
• |
Ocuphire might have to suspend or terminate clinical trials for various reasons, including a finding that the participants are being exposed to unacceptable health risks; |
• |
the cost of clinical trials may be greater than Ocuphire anticipates, including due to the ongoing COVID-19 pandemic or other public health emergency; |
• |
the supply or quality of any product candidate or other materials necessary to conduct clinical trials may be insufficient or inadequate; |
• |
the product candidate may have undesirable side effects or other unexpected characteristics, causing Ocuphire or its investigators, regulators or IRBs to suspend or terminate the trials; |
• |
clinical trials may be delayed or terminated because of the ongoing COVID-19 pandemic or another public health emergency; and |
• |
federal agencies may, due to reduced manpower or diverted resources to the COVID-19 pandemic, require more time to review clinical trial protocols and INDs. |
11
If Ocuphire experiences delays or difficulties in the enrollment of patients in clinical trials, Ocuphire’s ability to conduct and complete those clinical trials, and its ability to seek and receive necessary regulatory approvals, could be delayed or prevented.
Ocuphire or its future collaborators may not be able to initiate or continue clinical trials for its product candidates if Ocuphire is unable to locate and enroll a sufficient number of eligible patients to participate in these trials as required by the FDA or analogous regulatory authorities outside the United States. Patient enrollment can be affected by many factors, including:
• |
severity of the disease under investigation; |
• |
availability and efficacy of medications already approved for the disease under investigation; |
• |
eligibility criteria for the trial in question; |
• |
competition for eligible patients with other companies conducting clinical trials for product candidates seeking to treat the same indication or patient population; |
• |
its payments for conducting clinical trials; |
• |
perceived risks and benefits of the product candidate under study; |
• |
efforts to facilitate timely enrollment in clinical trials; |
• |
patient referral practices of physicians; |
• |
the ability to monitor patients adequately during and after treatment; |
• |
proximity and availability of clinical trial sites for prospective patients; |
• |
the ability of patients to safely participate in clinical trials during the COVID-19 pandemic or other public health emergencies; and |
• |
the ability to monitor patients adequately during periods in which social distancing is required or recommended due to the COVID-19 pandemic. |
Ocuphire expects that its late stage clinical trials of Nyxol and APX3330 will commence in the fourth quarter of 2020 through the first quarter of 2021 and each trial may take up to 3 to 9 months to enroll; however, Ocuphire cannot assure you that its timing and enrollment assumptions are correct given the above factors. The recent COVID-19 pandemic may also increase the time required to recruit patients for a study, and may also diminish the ability to monitor patients during the clinical trial. Ocuphire’s inability to enroll a sufficient number of patients for its clinical trials or retain sufficient enrollment through the completion of its trials would result in significant delays or may require Ocuphire to abandon one or more clinical trials altogether. Enrollment delays in Ocuphire’s clinical trials may result in increased development costs for its product candidates and cause its stock price to decline.
Ocuphire or others could discover that Ocuphire’s product candidates lack sufficient efficacy, or that they cause undesirable side effects that were not previously identified, which could delay or prevent regulatory approval or commercialization.
Because both Nyxol and APX3330 have been tested in relatively small patient populations, at a limited range of daily doses up to .75% Phentolamine Ophthalmic Solution (which is the same as 1% Phentolamine Mesylate Ophthalmic Solution) and 720 mg respectively, and for limited durations to date, it is possible that Ocuphire’s clinical trials have or will indicate an apparent positive effect of Nyxol or APX3330 that is greater than the actual positive effect, if any, or that additional and unforeseen side effects may be observed as its development progresses. Additionally, the combination product candidate of Nyxol and pilocarpine may not achieve the efficacy that is expected based on the individual contributions to efficacy. The discovery that either Nyxol or APX3330 lacks sufficient efficacy, or that they cause undesirable side effects (including side effects not previously identified in Ocuphire’s completed clinical trials), could cause Ocuphire or regulatory authorities to interrupt, delay, or discontinue clinical trials, and could result in the denial of regulatory approval by the FDA or other non-U.S. regulatory authorities for any or all targeted indications.
12
The discovery that Ocuphire’s product candidates lack sufficient efficacy or that they cause undesirable side effects that were not previously identified could prevent Ocuphire from commercializing such product candidates and generating revenues from sales. In addition, if Ocuphire receives marketing approval for its product candidates and Ocuphire or others later discover that it is less effective, or identify undesirable side effects caused by its product candidates:
• |
regulatory authorities may withdraw their approval of the product; |
• |
Ocuphire may be required to recall the product, change the way this product is administered, conduct additional clinical trials, or change the labeling or distribution of the product (including REMS); |
• |
additional restrictions may be imposed on the marketing of, or the manufacturing processes for, the product; |
• |
Ocuphire may be subject to fines, injunctions, or the imposition of civil or criminal penalties; |
• |
Ocuphire could be sued and held liable for harm caused to patients; |
• |
the product may be rendered less competitive and sales may decrease; or |
• |
Ocuphire’s reputation may suffer generally both among clinicians and patients. |
Any one or a combination of these events could prevent Ocuphire from achieving or maintaining market acceptance of the affected product candidate, or could substantially increase the costs and expenses of commercializing the product candidate, which in turn could delay or prevent Ocuphire from generating significant, or any, revenues from the sale of the product candidate.
Changes in regulatory requirements or FDA guidance, or unanticipated events during Ocuphire’s clinical trials, may result in changes to clinical trial protocols or additional clinical trial requirements, which could result in increased costs to Ocuphire or delays in its development timeline.
Changes in regulatory requirements or FDA guidance, or unanticipated events during Ocuphire’s clinical trials, may force Ocuphire to amend clinical trial protocols or the FDA may impose additional clinical trial requirements. Amendments to Ocuphire’s clinical trial protocols would require resubmission to the FDA and IRBs for review and approval, and may adversely impact the cost, timing or successful completion of a clinical trial. If Ocuphire experiences delays completing, or if it terminates, any Phase 2 or Phase 3 trials, or if it is required to conduct additional clinical trials, the commercial prospects for its product candidates may be harmed and its ability to generate product revenues will be delayed.
If Ocuphire fails to receive regulatory approval for any of its planned indications for its product candidates or fails to develop additional product candidates, Ocuphire’s commercial opportunity will be limited.
Ocuphire is initially focused on the development of its product candidates for its target indications, the treatment of NVD, pharmacologically-induced mydriasis, presbyopia, DR and DME. However, Ocuphire cannot assure you that it will be able to obtain regulatory approval of its product candidates for any indication, or successfully commercialize its product candidates, if approved. If Ocuphire does not receive regulatory approval for, or successfully commercialize, its product candidates for one or more of its targeted or other indications, Ocuphire’s commercial opportunity will be limited.
Ocuphire may pursue clinical development of additional acquired or in-licensing product candidates. Developing, obtaining regulatory approval for and commercializing additional product candidates will require substantial additional funding beyond the net proceeds of the Pre-Merger Financing, and are prone to the risks of failure inherent in drug product development. Ocuphire cannot assure you that it will be able to successfully advance any additional product candidates through the development process.
Even if it obtains FDA approval to market additional product candidates, Ocuphire cannot assure you that any such product candidates will be successfully commercialized, widely accepted in the marketplace, or more effective than other commercially available alternatives. If Ocuphire is unable to successfully develop and commercialize additional product candidates, its commercial opportunity will be limited.
Ocuphire has limited drug research and discovery capabilities and may need to acquire or license product candidates from third parties to expand its product candidate pipeline.
Ocuphire currently has limited drug research and discovery capabilities. Accordingly, if it is to expand its product candidate pipeline beyond Nyxol and APX3330 and its pipeline candidates, Ocuphire may need to acquire
13
or license product candidates from third parties. Ocuphire would face significant competition in seeking to acquire or license promising product candidates. Many of its competitors for such promising product candidates may have significantly greater financial resources and more extensive experience in preclinical testing and clinical trials, obtaining regulatory approvals, and manufacturing and marketing pharmaceutical products, and thus, may be a more attractive option to a potential licensor than Ocuphire. If Ocuphire is unable to acquire or license additional promising product candidates, it may not be able to expand its product candidate pipeline.
If Ocuphire is able to acquire or license other product candidates, such license agreements will likely impose various obligations upon it, and its licensors may have the right to terminate the license thereunder in the event of a material breach or, in some cases, at will. A termination of a future license could result in Ocuphire’s loss of the right to use the licensed intellectual property, which could adversely affect Ocuphire’s ability to develop and commercialize a future product candidate, if approved, as well as harm its competitive business position and its business prospects.
Ocuphire may expend its limited resources to pursue a particular indication and fail to capitalize on indications that may be more profitable or for which there is a greater likelihood of success.
Because Ocuphire has limited financial and managerial resources, it is currently focusing only on development programs that it identifies for specific indications for its product candidates. As a result, Ocuphire may forego or delay pursuit of opportunities for other indications, or with other potential product candidates that later prove to have greater commercial potential. Ocuphire’s resource allocation decisions may cause it to fail to capitalize on viable commercial products or profitable market opportunities. Ocuphire’s spending on current and future research and development programs for specific indications or future product candidates may not yield any commercially viable product. If Ocuphire does not accurately evaluate the commercial potential or target market for its product candidates, it may not gain approval or achieve market acceptance of that candidate, and its business and financial results will be harmed.
Risks Related to Ocuphire’s Financial Position and Need for Additional Capital
Ocuphire has incurred only losses since inception. Ocuphire expects to incur losses for the foreseeable future and may never achieve or maintain profitability.
Since inception, Ocuphire incurred only operating losses. Prior to the Merger, Ocuphire Private Company’s net losses were approximately $6.2 million and $1.6 million for the years ended December 31, 2019 and 2018, respectively, and $5.9 million for the nine-month period ended September 30, 2020. As of September 30, 2020, Ocuphire Private Company had an accumulated deficit of $14 million. Ocuphire has funded its operations primarily through issuance of common stock, warrants, promissory notes and convertible notes in private placements. It has devoted substantially all of its financial resources and efforts on research and development, including clinical development of its product candidates. Even assuming Ocuphire obtains regulatory approval for one or more of its product candidates, Ocuphire expects that it will be at least three years before it has a product candidate ready for commercialization. Ocuphire expects to continue to incur significant expenses and increased operating losses for the foreseeable future.
To become and remain profitable, Ocuphire must develop and eventually commercialize a product with market potential. This will require Ocuphire to be successful in a range of challenging activities, including completing preclinical testing and clinical trials, obtaining regulatory approval for a product candidate, manufacturing, marketing, and selling any drug for which it may obtain regulatory approval and satisfying any post-marketing requirements. Ocuphire is the early stages of most of these activities. Ocuphire may never succeed in these activities and, even if it does, it may never generate revenues that are significant or large enough to achieve profitability.
If Ocuphire does achieve profitability, it may not be able to sustain or increase profitability on an annual basis. Its failure to become or remain profitable may decrease Ocuphire’s value and could impair its ability to raise capital, maintain its research and development efforts, expand its business, or continue its operations.
14
Ocuphire has not generated any revenue and may never be profitable.
Ocuphire’s ability to become profitable depends upon its ability to generate revenue. To date, Ocuphire has not generated any revenue from its product candidates, Nyxol and APX3330, and it does not currently have any other products or product candidates. Ocuphire does not know if, or when, it will generate any revenue. Ocuphire does not expect to generate significant revenue unless and until it obtains marketing approval of, and commercializes, Nyxol or APX3330. Ocuphire’s ability to generate revenue depends on a number of factors, including its ability to:
• |
obtain favorable results from and complete the clinical development of both Nyxol and APX3330 for their planned indications, including successful completion of the Phase 2 and Phase 3 trials for these indications; |
• |
submit an application to regulatory authorities for both product candidates and receive marketing approval in the United States and foreign countries; |
• |
contract for the manufacture of commercial quantities of its product candidates at acceptable cost levels; |
• |
establish sales and marketing capabilities to effectively market and sell its product candidates in the United States or other markets, alone or with a pharmaceutical partner; and |
• |
achieve market acceptance of its product candidates in the medical community and with third-party payors. |
Even if Ocuphire’s product candidates are approved for commercial sale in one or all of the initial indications that it is pursuing, they may not gain market acceptance or achieve commercial success. In addition, Ocuphire anticipates incurring significant costs associated with commercializing its product candidates. Ocuphire may not achieve profitability soon after generating product revenue, if ever, and may be unable to continue operations without continued funding.
Ocuphire’s recurring operating losses have raised substantial doubt regarding its ability to continue as a going concern.
Ocuphire’s recurring operating losses raise substantial doubt about its ability to continue as a going concern. For the fiscal year ended December 31, 2019, its independent registered public accounting firm has issued its report on Ocuphire’s financial statements and has expressed substantial doubt about its ability to continue as a going concern. Ocuphire has no current source of revenue to sustain its present activities, and it does not expect to generate revenue until and unless the FDA or other applicable regulatory authorities approves, and it successfully commercializes, its product candidates. Accordingly, Ocuphire’s ability to continue as a going concern will require it to obtain additional financing to fund its operations. Uncertainty surrounding Ocuphire’s ability to continue as a going concern may make it more difficult for it to obtain financing for the continuation of its operations and could result in a loss of confidence by investors, suppliers, contractors, and employees.
Ocuphire’s relatively short operating history may make it difficult for investors to evaluate the success of its business to date and to assess its future viability.
Ocuphire is a clinical-stage company, and its operations to date have been limited to organizing and staffing its company, business planning, raising capital, and developing its product candidates. Ocuphire has not yet demonstrated its ability to successfully complete a Phase 3 program, obtain regulatory approval, manufacture a product at commercial scale, or conduct sales and marketing activities necessary for successful product commercialization.
Additionally, there is no operating history on which you may evaluate this business and its prospects. Investment in a start-up company such as Ocuphire is inherently subject to many risks. These risks and difficulties include challenges in accurate financial planning as a result of: (a) accumulated losses; (b) uncertainties resulting from a relatively limited time period in which to develop and evaluate business strategies as compared to companies with longer operating histories; (c) compliance with regulation required to commence sales on some future products; (d) reliance on third parties for operations; (e) financing the business; and (f) meeting the challenges of the other risk factors described herein. Ocuphire has no operating history upon which investors may base an evaluation of its performance; therefore, it is subject to all risks incident to the creation and development of a new business. There can be no assurance that Ocuphire can realize its plans on the projected timetable in order to reach sustainable or profitable operations.
15
Ocuphire will need substantial additional capital in the future. If additional capital is not available, it will have to delay, reduce or cease operations.
Although Ocuphire believes that the net proceeds from the Pre-Merger Financing, together with cash on hand, will be sufficient to fund its operations through 2021, Ocuphire will need to raise additional capital to continue to fund the further development of its product candidates and operations. Its future capital requirements may be substantial and will depend on many factors including:
• |
the scope, size, rate of progress, results, and costs of researching and developing its product candidates, and initiating and completing its preclinical studies and clinical trials; |
• |
the cost, timing and outcome of its efforts to obtain marketing approval for its product candidates in the United States and other countries, including to fund the preparation and filing of an NDA with the FDA for its product candidates and to satisfy related FDA requirements and regulatory requirements in other countries; |
• |
the number and characteristics of any additional product candidates it develops or acquires, if any; |
• |
Ocuphire’s ability to establish and maintain collaborations on favorable terms, if at all; |
• |
the amount of revenue, if any, from commercial sales, should its product candidates receive marketing approval; |
• |
the costs associated with commercializing its product candidates, if Ocuphire receives marketing approval, including the cost and timing of developing sales and marketing capabilities or entering into strategic collaborations to market and sell its product candidates; |
• |
the cost of manufacturing its product candidates or products Ocuphire successfully commercializes; and |
• |
the costs associated with general corporate activities, such as the cost of filing, prosecuting and enforcing patent claims and making regulatory filings. |
Changing circumstances may cause Ocuphire to consume capital significantly faster than it currently anticipates. Because the outcome of any clinical trial is highly uncertain, Ocuphire cannot reasonably estimate the actual amounts necessary to successfully complete the development, regulatory approval and commercialization of its product candidates. Additional financing may not be available when Ocuphire needs it, or may not be available on terms that are favorable to Ocuphire. In addition, Ocuphire may seek additional capital due to favorable market conditions or strategic considerations, even if Ocuphire believes it has sufficient funds for its current or future operating plans. If adequate funds are unavailable to it on a timely basis, or at all, Ocuphire may not be able to continue the development its product candidates, or commercialize its product candidates, if approved, unless it finds a strategic partner.
Raising additional capital may cause dilution to Ocuphire’s stockholders, restrict Ocuphire’s operations, or require Ocuphire to relinquish rights to its technologies or product candidates.
Until such time, if ever, as Ocuphire can generate substantial product revenues, it expects to finance its cash needs through a combination of equity and debt financings as well as potential strategic collaborations and licensing arrangements. It does not have any committed external source of funds. Debt financing or preferred equity financing, if available, may involve agreements that include covenants limiting or restricting Ocuphire’s ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends.
If Ocuphire raises funds through strategic collaborations or marketing, distribution, or licensing arrangements with third parties, it may have to relinquish valuable rights to its technologies, future revenue streams, research programs or product candidates or to grant licenses on terms that may not be favorable to it. If it is unable to raise additional funds when needed, Ocuphire may be required to delay, limit, reduce or terminate its product development or future commercialization efforts or grant rights to develop and market product candidates that it would otherwise prefer to develop and market itself. This may reduce the value of its common stock.
16
Risks Related to Government Regulation
The FDA requires the completion of a toxicology study of similar duration before trials longer than 6 months can be conducted such as Phase 3 safety exposure trials for chronic indications or efficacy trials with such 6 month endpoints. This may lead to a significant delay in the commencement of long term clinical trials by Ocuphire or the failure of its product candidates to obtain marketing approval.
At this time, Ocuphire can run trials using Nyxol up to 28 days in duration based on its completed 28-day rabbit toxicology study. Therefore, the planned Phase 3 registration efficacy trials for NVD, with dosing for 7 to 14 days, may be conducted without further toxicology studies. Until Ocuphire has completed a six-month toxicology for Nyxol, FDA regulations restrict it from conducting clinical trials of six months or more in duration targeting chronic indications, which at this time is only the planned 1 year Phase 3 safety exposure trial for NVD. Ocuphire plans to initiate the in-life portion of the six-month toxicology study in rabbits for Nyxol in the first quarter of 2021, with an expected completion and draft report 12 months later. For APX3330, the drug has already been dosed for more than a year in humans and completed over 15 single- and repeat-dose toxicology studies in rats and dogs (including 2 studies up to 3 months in duration); with this data the FDA has reviewed, with no comments, Ocuphire’s planned 24 week clinical trial without the need for further toxicology studies needed. However, the FDA may require Ocuphire to complete further animal toxicology studies for future clinical trials prior to any marketing approval from regulatory authorities for the sale of APX3330. Clinical trials may be delayed due to these clinical restrictions and additional oversight by the FDA. In addition, the findings in the toxicology studies could impact the NDA reviews, and, if approved, labels and uses of Ocuphire’s product candidates.
Even if it receives marketing approval for its product candidates in the United States, Ocuphire may never receive regulatory approval to market such product candidates outside of the United States.
In addition to the United States, Ocuphire intends to seek regulatory approval to market its product candidates in Europe, Japan, Canada, and Australia, and potentially other markets. If Ocuphire pursues additional product candidates in the future, it may seek regulatory approval of such product candidates outside the United States. In order to market any product outside of the United States, however, Ocuphire must establish and comply with the numerous and varying safety, efficacy and other regulatory requirements of these other countries. Approval procedures vary among countries and can involve additional product candidate testing and additional administrative review periods. The time required to obtain approvals in other countries might differ from that required to obtain FDA approval. The marketing approval processes in other countries may include all of the risks detailed above regarding FDA approval in the United States as well as other risks. In particular, in many countries outside of the United States, products must receive pricing and reimbursement approval before the product can be commercialized. Obtaining this approval can result in substantial delays in bringing products to market in such countries. Marketing approval in one country does not ensure marketing approval in another, but a failure or delay in obtaining marketing approval in one country may have a negative effect on the regulatory process in others. Failure to obtain marketing approval in other countries or any delay or other setback in obtaining such approval would impair Ocuphire’s ability to market its product candidates in such foreign markets. Any such impairment would reduce the size of Ocuphire’s potential market, which could have an adverse impact on its business, results of operations and prospects.
Even if Ocuphire obtains marketing approval for its product candidates, such product candidates could be subject to post-marketing restrictions or withdrawal from the market, and Ocuphire may be subject to substantial penalties if it fails to comply with regulatory requirements or experience unanticipated problems with a product following approval.
Any product candidate for which Ocuphire, or its future collaborators, obtains marketing approval in the future, as well as the manufacturing processes, post-approval studies and measures, labeling, advertising, and promotional activities for such drug, among other things, will be subject to continual requirements of and review by the FDA and other regulatory authorities. These requirements include submissions of safety and other post-marketing information and reports, registration and listing requirements, requirements relating to manufacturing, quality control, quality assurance and corresponding maintenance of records and documents, requirements regarding the distribution of samples to physicians and recordkeeping. Even if marketing approval of a product candidate is granted, the approval may be subject to limitations on the indicated uses for which the drug may be marketed or to the conditions of approval, including the requirement to implement a REMS, which could include requirements for a restricted distribution system.
The FDA may also impose requirements for costly post-marketing studies or clinical trials and surveillance to monitor the safety or efficacy of a product candidate. The FDA and other agencies, including the Department of
17
Justice, closely regulate and monitor the post-approval marketing and promotion of drugs to ensure that they are manufactured, marketed, and distributed only for the approved indications and in accordance with the provisions of the approved labeling. The FDA imposes stringent restrictions on manufacturers’ communications regarding off-label use and if Ocuphire, or any future collaborator, does not market a product candidate for which it receives marketing approval for only its approved indications, Ocuphire, or the collaborator, may be subject to warnings or enforcement action for off-label promotion. Violation of the Federal Food, Drug, and Cosmetic Act (“FDC Act”) and other statutes, including the False Claims Act, relating to the promotion and advertising of prescription drugs, may lead to investigations or allegations of violations of federal or state healthcare fraud and abuse laws and state consumer protection laws.
In addition, later discovery of previously unknown AEs or other problems with Ocuphire’s product candidates or its manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may yield various results, including:
• |
litigation involving patients taking Ocuphire’s drugs; |
• |
restrictions on such drugs, manufacturers, or manufacturing processes; |
• |
restrictions on the labeling or marketing of a drug; |
• |
restrictions on drug distribution or use; |
• |
requirements to conduct post-marketing studies or clinical trials; |
• |
warning letters or untitled letters; |
• |
withdrawal of the drugs from the market; |
• |
refusal to approve pending applications or supplements to approved applications that Ocuphire submits; |
• |
product recall or public notification or medical product safety alerts to healthcare professionals; |
• |
fines, restitution, or disgorgement of profits or revenues; |
• |
suspension or withdrawal of marketing approvals; |
• |
damage to relationships with any potential collaborators; |
• |
unfavorable press coverage and damage to Ocuphire’s reputation; |
• |
refusal to permit the import or export of drugs; |
• |
product seizure; or |
• |
injunctions or the imposition of civil or criminal penalties. |
Ocuphire may seek to avail itself of mechanisms to expedite the development or approval for product candidates it may pursue in the future, such as fast track or breakthrough designation, but such mechanisms may not actually lead to a faster development or regulatory review or approval process.
Ocuphire may seek fast track designation, breakthrough designation, orphan drug designation, priority review, or accelerated approval for product candidates it may pursue in the future. For example, if a drug is intended for the treatment of a serious or life-threatening condition and the drug demonstrates the potential to address unmet medical needs for this condition, the drug sponsor may apply for FDA fast track designation. However, the FDA has broad discretion with regard to these mechanisms, and even if Ocuphire believes a particular product candidate is eligible for any such mechanism, it cannot guarantee that the FDA would decide to grant it. Even if it does obtain fast track or priority review designation or pursue an accelerated approval pathway, Ocuphire may not experience a faster development process, review, or approval compared to conventional FDA procedures. The FDA may withdraw a particular designation if it believes that the designation is no longer supported by data from Ocuphire’s clinical development program.
A breakthrough therapy is defined as a drug that is intended, alone or in combination with one or more other drugs, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints. Designation as a breakthrough therapy is within the discretion of the FDA. Accordingly, even if Ocuphire believes a product candidate meets the criteria for designation as a breakthrough therapy, the FDA may disagree and
18
instead determine not to make such designation. Ocuphire cannot be sure that its evaluation of a product candidate as qualifying for breakthrough therapy designation will meet the FDA’s requirements. In any event, the receipt of a breakthrough therapy designation for a product candidate may not result in a faster development process, review, or approval compared to conventional FDA procedures and does not assure ultimate approval by the FDA. In addition, even if one or more product candidates qualifies as a breakthrough therapy, the FDA may later decide that the product candidate no longer meets the conditions for qualification or may decide that the time period for FDA review or approval will not be shortened.
Recently enacted and future legislation may increase the difficulty and cost for Ocuphire and its future collaborators to obtain marketing approval of its product candidates and affect their pricing.
In the United States and some foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of a product candidate, restrict or regulate post-approval activities and affect Ocuphire’s ability, or the ability of its future collaborators, to profitably sell any drug for which it, or they, obtains marketing approval. Ocuphire expects that current laws, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and cause downward pressure on the price that Ocuphire, or its future collaborators, may charge for any approved drug.
For example, in March 2010, the United States Congress enacted the Patient Protection and Affordable Care Act (“PPACA”) and the Health Care and Education Reconciliation Act, or the Healthcare Reform Act, which expanded health care coverage through Medicaid expansion and the implementation of the individual mandate for health insurance coverage and which included changes to the coverage and reimbursement of drug products under government healthcare programs. Under the Trump administration, there have been ongoing efforts to modify or repeal all or certain provisions of the Healthcare Reform Act. For example, tax reform legislation was enacted at the end of 2017 that eliminates the tax penalty established under Healthcare Reform Act for individuals who do not maintain mandated health insurance coverage beginning in 2019. The Healthcare Reform Act has also been subject to judicial challenge. In December 2018, a federal district court, in a challenge brought by a number of state attorneys general, found the Healthcare Reform Act unconstitutional in its entirety because, once Congress repealed the individual mandate provision, there was no longer a basis to rely on Congressional taxing authority to support enactment of the law. Pending appeals, which could take some time, the Healthcare Reform Act is still operational in all respects.
There have also been other reform initiatives under the Trump Administration, including initiatives focused on drug pricing. For example, the Bipartisan Budget Act of 2018 contained various provisions that affect coverage and reimbursement of drugs, including an increase in the discount that manufacturers of Medicare Part D brand name drugs must provide to Medicare Part D beneficiaries during the coverage gap from 50% to 70% that took effect in 2019. As another example, in May of 2018, President Trump and the Secretary of the Department of Health and Human Services, or HHS, released a “blueprint” to lower prescription drug prices and out-of-pocket costs. Certain proposals in the blueprint, and related drug pricing measures proposed since the blueprint, could cause significant operational and reimbursement changes for the pharmaceutical industry. As another example, in November of 2018, CMS issued an advance notice of proposed rulemaking that proposed revisions to Medicare Part D to support health plans’ negotiation of lower drug prices with manufacturers and reduce health plan members’ out-of-pocket costs.
There have also been efforts by federal and state government officials or legislators to implement measures to regulate prices or payment for pharmaceutical products, including legislation on drug importation. Recently, there has been considerable public and government scrutiny of pharmaceutical pricing and proposals to address the perceived high cost of pharmaceuticals. There have also been recent state legislative efforts to address drug costs, which generally have focused on increasing transparency around drug costs or limiting drug prices.
General legislative cost control measures may also affect reimbursement for Ocuphire’s product candidates. The Budget Control Act, as amended, resulted in the imposition of 2% reductions in Medicare (but not Medicaid) payments to providers in 2013 and will remain in effect through 2027 unless additional Congressional action is taken. Any significant spending reductions affecting Medicare, Medicaid or other publicly funded or subsidized health programs that may be implemented and/or any significant taxes or fees that may be imposed on Ocuphire could have an adverse impact on results of operations.
Adoption of new legislation at the federal or state level could affect demand for, or pricing of, Ocuphire’s current or future products if approved for sale. Ocuphire cannot, however, predict the ultimate content, timing or effect of
19
any changes to the Healthcare Reform Act or other federal and state reform efforts. There is no assurance that federal or state health care reform will not adversely affect Ocuphire’s future business and financial results.
There have been judicial and congressional challenges and amendments to certain aspects of the PPACA, and Ocuphire expects there will be additional challenges and amendments to the PPACA in the future, as well as efforts to repeal and replace it. In addition, other legislative changes have been proposed and adopted since the PPACA was enacted. These new laws have resulted in additional reductions in Medicare and other healthcare funding and otherwise may affect the prices Ocuphire may obtain for any product candidate for which marketing approval is obtained. Any reduction in reimbursement from Medicare or other government-funded programs may result in a similar reduction in payments from private payors. Moreover, recently there has been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products. The implementation of cost containment measures or other healthcare reforms may prevent Ocuphire from being able to generate revenue, attain profitability, or commercialize its drugs.
Legislative and regulatory proposals have been made to expand post-approval requirements and restrict sales and promotional activities for pharmaceutical products. Ocuphire cannot be sure whether additional legislative changes will be enacted, or whether the FDA regulations, guidance, or interpretations will be changed, or what the impact of such changes on the marketing approvals of a product candidate, if any, may be. In addition, increased scrutiny by the U.S. Congress of the FDA’s approval process may significantly delay or prevent marketing approval, or subject Ocuphire or its future collaborators to more stringent drug labeling and post-marketing testing and other requirements.
Governments outside of the United States tend to impose strict price controls, which may adversely affect Ocuphire’s revenues from the sales of a drug, if any.
In some countries, particularly in the European Union, the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after the receipt of marketing approval for a drug. To obtain reimbursement or pricing approval in some countries, Ocuphire, or its future collaborators, may be required to conduct a clinical trial that compares the cost-effectiveness of its products to other available therapies. If reimbursement of Ocuphire’s drugs are unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, its business could be harmed.
Ocuphire’s relationships with healthcare providers and third-party payors will be subject to applicable fraud and abuse and other healthcare laws and regulations, which could expose Ocuphire to criminal sanctions, civil penalties, contractual damages, reputational harm, and diminished profits and future earnings, among other penalties and consequences.
Healthcare providers and third-party payors will play a primary role in the recommendation and prescription of any product candidate for which Ocuphire obtains marketing approval. Ocuphire’s current and future arrangements with third-party payors and customers may expose Ocuphire to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements and relationships through which it markets, sells, and distributes product candidates for which it obtains marketing approval. Restrictions and obligations under applicable federal and state healthcare laws and regulations include the following:
• |
the federal Anti-Kickback Statute prohibits, among other things, persons and entities from knowingly and willfully soliciting, offering, receiving or providing remuneration, directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made under a federal healthcare program such as Medicare and Medicaid; |
• |
the federal false claims and civil monetary penalties laws, including the civil False Claims Act, impose criminal and civil penalties, including civil whistleblower or qui tam actions, against individuals or entities for knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or fraudulent or making a false statement to avoid, decrease, or conceal an obligation to pay money to the federal government; |
• |
HIPAA imposes criminal and civil liability for, among other things, executing a scheme to defraud any healthcare benefit program or making false statements relating to healthcare matters; |
20
• |
HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, and its implementing regulations, also imposes obligations, including mandatory contractual terms, on certain people and entities with respect to safeguarding the privacy, security, and transmission of individually identifiable health information; |
• |
the federal Physician Payments Sunshine Act under the Affordable Care Act requires certain manufacturers of drugs, devices, biologics, and medical supplies for which payment is available under Medicare, Medicaid, or the Children’s Health Insurance Program, with specific exceptions, to report specially to the Centers for Medicare & Medicaid Services within the U.S. Department of Health and Human Services information related to physician payments and other transfers of value and physician ownership and investment interests; and |
• |
analogous state and foreign laws and regulations, such as state anti-kickback and false claims laws, may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third-party payors, including private insurers, and some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government, in addition to requiring drug manufacturers to report information related to payments to physicians and other healthcare providers or marketing expenditures. Certain state and foreign laws also govern the privacy and security of health information in ways that differ from each other and often are not preempted by HIPAA, thus complicating compliance efforts. |
Efforts to ensure that Ocuphire’s current and future business arrangements with third parties will comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that Ocuphire’s business practices may not comply with current or future statutes, regulations, or case law involving applicable fraud and abuse or other healthcare laws and regulations. If Ocuphire’s operations are found to be in violation of any of these laws or any other governmental regulations that may apply to it, it may be subject to significant civil, criminal, and administrative penalties, damages, fines, exclusion from government funded healthcare programs, such as Medicare and Medicaid, disgorgement, individual imprisonment, contractual damages, reputational harm, diminished profits and future earnings, and the curtailment or restructuring of its operations. If any of the physicians or other providers or entities with whom Ocuphire expects to do business are found to not be in compliance with applicable laws, they may be subject to criminal, civil, and administrative sanctions, including exclusions from government funded healthcare programs. Defending against any such actions can be costly, time-consuming, and may require significant financial and personnel resources. Therefore, even if Ocuphire is successful in defending against any such actions that may be brought against it, its business may be impaired.
Ocuphire is subject to U.S. and certain foreign export and import controls, sanctions, embargoes, anti-corruption laws, and anti-money laundering laws and regulations. Compliance with these legal standards could impair its ability to compete in domestic and international markets. Ocuphire could face criminal liability and other serious consequences for violations which could harm its business.
Ocuphire is subject to export control and import laws and regulations, including the U.S. Export Administration Regulations, U.S. Customs regulations, various economic and trade sanctions regulations administered by the U.S. Treasury Department’s Office of Foreign Assets Controls, the U.S. Foreign Corrupt Practices Act of 1977, as amended, the U.S. domestic bribery statute contained in 18 U.S.C. § 201, the U.S. Travel Act, the USA PATRIOT Act, and other state and national anti-bribery and anti-money laundering laws in the countries in which Ocuphire conducts activities. Anti-corruption laws are interpreted broadly and prohibit companies and their employees, agents, contractors, and other partners from authorizing, promising, offering, or providing, directly or indirectly, improper payments or anything else of value to recipients in the public or private sector. Ocuphire may engage third parties for clinical trials outside of the United States, to sell its products abroad once it enters a commercialization phase, and/or to obtain necessary permits, licenses, patent registrations, and other regulatory approvals. Ocuphire has direct or indirect interactions with officials and employees of government agencies or government-affiliated hospitals, universities, and other organizations. Ocuphire can be held liable for the corrupt or other illegal activities of its employees, agents, contractors, and other partners, even if it does not explicitly authorize or have actual knowledge of such activities. Any violations of the laws and regulations described above may result in substantial civil and criminal fines and penalties, imprisonment, the loss of export or import privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm, and other consequences.
21
Ocuphire employees may engage in misconduct or other improper activities, including violating applicable regulatory standards and requirements or engaging in insider trading, which could significantly harm Ocuphire’s business.
Ocuphire is exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include intentional failures to:
• |
comply with the regulations of the FDA and applicable non-U.S. regulators; |
• |
provide accurate information to the FDA and applicable non-U.S. regulators; |
• |
comply with healthcare fraud and abuse laws and regulations in the United States and abroad; |
• |
report financial information or data accurately; or |
• |
disclose unauthorized activities to Ocuphire. |
In particular, sales, marketing, and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, misconduct, kickbacks, self-dealing, and other abusive practices. These laws and regulations restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs, and other business arrangements. Employee misconduct could also involve the improper use of, including trading on, information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to Ocuphire’s reputation. It is not always possible to identify and deter employee misconduct, and the precautions Ocuphire takes to detect and prevent this activity may be ineffective in controlling unknown or unmanaged risks or losses or in protecting it from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against Ocuphire, and Ocuphire is not successful in defending itself or asserting its rights, those actions could have a significant impact on its business, including the imposition of significant civil, criminal, and administrative penalties, damages, fines, exclusion from government funded healthcare programs such as Medicare and Medicaid, disgorgement, individual imprisonment, contractual damages, reputational harm, diminished profits and future earnings, and the curtailment or restructuring of its operations.
The FDA and other regulatory agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses. If found to have improperly promoted off-label uses, Ocuphire may become subject to significant liability.
The FDA and other regulatory agencies strictly regulate the promotional claims that may be made about prescription products. In particular, a product may not be promoted for uses that are not approved by the FDA or such other regulatory agencies as reflected in the product’s approved labeling. If Ocuphire receives marketing approval for its product candidates for a certain indication, physicians may nevertheless prescribe such products to their patients in a manner that is inconsistent with the approved label. If Ocuphire is found to have promoted such off-label uses, it may become subject to significant liability. The federal government has levied large civil and criminal fines against companies for improper promotion and has enjoined several companies from engaging in off-label promotion. The FDA has also requested that companies enter into consent decrees or permanent injunctions under which specified promotional conduct is changed or curtailed. If Ocuphire cannot successfully manage the promotion of its product candidates, if approved, it could become subject to significant liability, which would adversely affect its business and financial condition.
Risks Related to Commercialization of Ocuphire’s Product Candidates
Ocuphire faces substantial competition, which may result in others discovering, developing, or commercializing products before or more successfully than it does.
The development and commercialization of new drug products is highly competitive. Ocuphire expects to face competition with respect to its product candidates, if approved, and will face competition with respect to any future product candidates that it may seek to develop or commercialize from major pharmaceutical companies, specialty pharmaceutical companies, biotechnology companies, universities and other research institutions, and government agencies worldwide. The ophthalmic therapies market is highly competitive and dynamic. Ocuphire’s success will depend, in part, on its ability to obtain a share of the market for its planned indications.
22
Nyxol
Ocuphire is developing Nyxol for use in three different indications: the treatment of NVD, the reversal of pharmacologically induced mydriasis (“RM”), and the treatment of presbyopia. In addition to currently approved therapies, any product that is developed for any of the three indications could compete with Nyxol. Such a product could reduce the overall market opportunity for Nyxol. Other pharmaceutical companies may develop therapies for the same indications that would compete with Nyxol, if approved, and that would not infringe the claims of Ocuphire’s patents, pending patent applications, or other proprietary rights, which could adversely affect its business and results of operations.
Currently, there are no available and approved pharmacological therapies for NVD or RM and Ocuphire is not aware of any in development. Rev-Eyes® (dapiprazole), an alpha-1 antagonist, was approved by the FDA in 1990 to reverse mydriasis induced by adrenergic or anticholinergic agents. Rev-Eyes was withdrawn in the past from the market for reasons unrelated to safety or efficacy, according to the FDA.
Presbyopia
There are currently no approved pharmacological treatments for presbyopia, though several drug treatments are in development. Currently, the competition includes reading glasses, multifocal contact lenses, and monovision contact lenses (i.e., where one eye wears a near vision lens and the other eye wears a distance vision lens). Ocuphire will also compete against several pharmacological therapies in development for the temporary treatment of presbyopia, some of which are pilocarpine-based pupil management therapies, including:
• |
Presbysol® (AGN-190584), with 1.25% pilocarpine, developed by Allergan plc. |
• |
Presbidrops® (CSF-1), with low dose pilocarpine and a secondary agent (lubricant), developed by Orasis Pharmaceuticals Ltd. |
• |
Liquid Vision®, with aceclidine (another miotic agent), developed by Presbyopia Therapies, LLC. |
• |
MicroLine®, which is a microdose formulation of pilocarpine, developed by Eyenovia, Inc. |
• |
KT-101, which uses pilocarpine in the AcuStream delivery system, developed by Kedalion Therapeutics, Inc. |
• |
BrimocholTM, with brimonidine and carbachol (both are miotic agents), developed by Visus Therapeutics, Inc. |
• |
UNR844, which uses a mechanism that involves softening the lens to increase near visual acuity, developed by Novartis AG (originally Encore Vision, Inc.). |
There are approved devices for presbyopia. One of these is the KAMRA Inlay, developed by AcuFocus, Inc. and marketed by SightLife Surgical, Inc. Another is the Eyelike NoanPinhole, developed by Koryo Eyetech, the first commercially available pinhole soft contact lens. Nyxol would not directly compete against these devices, but rather would be a non-invasive alternative for presbyopes who are averse to surgical intervention.
Glaucoma
Ocuphire may work with a partner to develop a combination approach with Nyxol and Latanoprost as a potential treatment strategy for glaucoma patients, and would face substantial competition. Glaucoma has many approved generic and prescription drug and non-drug treatments including: rho kinase inhibitors Rhopressa® and Rocklatan®, marketed by Aerie Pharmaceuticals, Inc.; latanoprostene bunod Vyzulta®, marketed by Bausch + Lomb, Inc.; prostaglandin analogues (“PGAs”), such as latanoprost; beta blockers, such as timolol; alpha agonists, such as brimonidine; carbonic anhydrase inhibitors, such as dorzolamide hydrochloride; cholinergic agonists, such as pilocarpine; combination therapies, such as Combigan®, marketed by Allergan, Inc., which combines brimonidine and timolol; and minimally invasive glaucoma surgery (“MIGS”).
APX3330
Ocuphire is developing APX3330 for use in two different indications initially: the treatment of DR and DME, and potentially later the treatment of wAMD. In addition to currently approved therapies, any product that is developed for either of the three indications could directly compete directly with APX3330. Such a product could reduce the overall market opportunity for APX3330. Other pharmaceutical companies may develop therapies for the
23
same indications that would compete with APX3330, if approved, and that would not infringe the claims of Ocuphire’s in-licensed patents, pending patent applications, or other proprietary rights, which could adversely affect its business and results of operations.
Competition in Diabetic Retinopathy / Diabetic Macular Edema / wAMD
Ocuphire may face potential competition from both existing therapies and those in development. Current therapies for these retinal diseases rely on suppressing VEGF activity via intravitreal injection or by mitigating the inflammation via intravitreal corticosteroid-releasing implants including:
• |
Lucentis® (ranibizumab) and Avastin® (bevacizumab), which are anti-VEGF monoclonal antibody intravitreal injections, developed by Genentech, Inc. |
• |
EYLEA® (aflibercept), a VEGF inhibitor intravitreal injection, developed by Regeneron Pharmaceuticals. |
• |
Beovu® Brolucizumab, an anti-VEGF monoclonal antibody intravitreal injection, developed by Novartis AG. |
• |
MACUGEN® (pegaptanib sodium injection), a selective inhibitor of VEGF-165, developed by Bausch + Lomb. |
• |
Ozurdex® (dexamethasone), a corticosteroid IVT implant, developed by Allergan plc. |
• |
Iluvien (fluocinolone acetonide), a corticosteroid IVT implant, developed by Alimera Sciences, Inc. |
• |
There are also several pharmacological therapies in development, including: |
• |
Abicipar, an anti-VEGF intravitreal injection with a long duration of action, developed by Allergan plc and Molecular Partners. |
• |
Farcimab, a bispecific antibody intravitreal injection that suppresses both VEGF and Angiopoietin-2, developed by Genentech, Inc. and Roche AG. |
• |
KSI-301, an anti-VEGF antibody intravitreal injection coupled with a biopolymer that is intended to increase the time between injections, developed by Kodiak Sciences. |
• |
OPT-302, an intravitreal injection which binds to multiple types of VEGF receptors that could be used with other anti-VEGF agents, developed by Opthea Limited. |
• |
ALG-1001, an integrin peptide therapy intravitreal injection that is being evaluated as a sequential or in-combination therapy with bevacizumab in patients with DME, developed by Allegro Ophthalmics, LLC. |
Ocuphire’s competitors may develop products that are more effective, safer, more convenient, or less costly than any that it is developing, or that would render its product candidates obsolete or non-competitive. Ocuphire’s competitors may also render its technologies obsolete by advances in existing technological approaches or the development of new or different approaches, potentially eliminating the advantages in Ocuphire’s drug discovery process. Ocuphire’s competitors may also obtain marketing approval from the FDA or other regulatory authorities for its products more rapidly than Ocuphire obtains approval for its products, which could result in Ocuphire’s competitors establishing a strong market position before Ocuphire is able to enter the market.
Many of Ocuphire’s competitors have significantly greater name recognition, financial resources, and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals, and marketing approved products than Ocuphire does. Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of Ocuphire’s competitors. Smaller and other early stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These companies compete with Ocuphire in recruiting, hiring, and retaining qualified scientific and management personnel, engaging contract service providers, manufacturers and consultants, establishing clinical trial sites, recruiting patients for clinical trials, and entering into strategic transactions, as well as in acquiring technologies complementary to, or necessary for, Ocuphire’s programs.
Ocuphire lacks experience in commercializing products, which may have an adverse effect on its business.
If its product candidates receive marketing approval, Ocuphire will need to transition from a company with a development focus to a company capable of supporting commercial activities, and it may not be successful in making
24
that transition. Ocuphire has never filed an NDA, and has not yet demonstrated the ability to obtain marketing approval for, or to commercialize, any product candidate. As a result, its clinical development and regulatory approval activities, and its ability to successfully commercialize any approved products, may involve more inherent risk, take longer, and cost more than would be the case if it were a company with experience obtaining marketing approval for and commercializing a product candidate.
If Ocuphire is unable to establish sales and marketing capabilities or enter into agreements with third parties to sell, market, and distribute its product candidates, if approved, it may not be successful in commercializing such product candidates if and when they are approved.
Ocuphire does not have any sales or marketing infrastructure and have no capabilities in place at the present time for the sale, marketing, or distribution of pharmaceutical products. To achieve commercial success for any approved product for which it retains sales and marketing responsibilities, Ocuphire must either develop a sales and marketing organization or outsource part or all of these functions to other third parties.
There are risks involved with Ocuphire both establishing its own sales and marketing capabilities and entering into arrangements with third parties to perform these services. For example, recruiting and training a sales force is expensive and time-consuming, which could delay any product launch. If the commercial launch of a product candidate for which Ocuphire recruits a sales force and establish marketing capabilities is delayed or does not occur for any reason, it would have prematurely or unnecessarily incurred these commercialization expenses. This may be costly, and Ocuphire’s investment would be lost if it cannot retain or reposition its sales and marketing personnel.
Factors that may inhibit Ocuphire’s efforts to commercialize its product candidates on its own include:
• |
the inability to recruit and retain adequate numbers of effective sales and marketing personnel or enter into distribution agreements with third parties; |
• |
the inability of sales personnel to obtain access to physicians or persuade adequate numbers of physicians to prescribe its product candidate; |
• |
the lack of complementary products to be offered by sales personnel, which may put Ocuphire at a competitive disadvantage relative to companies with more extensive product lines; |
• |
unforeseen costs and expenses associated with creating an independent sales and marketing organization; and |
• |
the inability to obtain sufficient coverage and reimbursement from third-party payors and governmental agencies. |
If it enters into arrangements with third parties to perform sales, marketing, and distribution services, Ocuphire’s product revenues or the profitability of these product revenues to it are likely to be lower than if it were to market and sell a product that Ocuphire developed itself. In addition, Ocuphire may not be successful in entering into arrangements with third parties to sell and market any product candidate or may be unable to do so on terms that are favorable to it. Ocuphire likely will have little control over such third parties, and any of them may fail to devote the necessary resources and attention to sell and market a drug effectively. If Ocuphire does not establish sales and marketing capabilities successfully, either on its own or in collaboration with third parties, it will not be successful in commercializing its product candidates.
Ocuphire’s future commercial success depends upon attaining significant market acceptance of its product candidates, if approved, among physicians, patients, third-party payors, and others in the medical community.
Even if Ocuphire’s product candidates receive marketing approval, they may nonetheless fail to gain sufficient market acceptance by physicians, patients, healthcare payors, or others in the medical community. If such product candidates do not achieve an adequate level of acceptance, Ocuphire may not generate significant product revenues and may not become profitable. The degree of market acceptance of a product candidate, if approved for commercial sale, will depend on a number of factors, including:
• |
efficacy and potential advantages compared to alternative treatments; |
• |
the ability to offer Ocuphire’s product for sale at competitive prices; |
• |
the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
25
• |
any restrictions on the use of Ocuphire’s product together with other medications; |
• |
interactions of its product with other medicines patients are taking; |
• |
inability of certain types of patients to take Ocuphire’s product; |
• |
demonstrated ability to treat patients and, if required by any applicable regulatory authority in connection with the approval for target indications as compared with other available therapies; |
• |
the relative convenience and ease of administration as compared with other treatments available for approved indications; |
• |
the prevalence and severity of any adverse side effects; |
• |
limitations or warnings contained in the labeling approved by the FDA; |
• |
availability of alternative treatments already approved or expected to be commercially launched in the near future; |
• |
the effectiveness of Ocuphire’s sales and marketing strategies; |
• |
Ocuphire’s ability to increase awareness through marketing efforts; |
• |
guidelines and recommendations of organizations involved in research, treatment and prevention of various diseases that may advocate for alternative therapies; |
• |
Ocuphire’s ability to obtain sufficient third-party coverage and adequate reimbursement; |
• |
the willingness of patients to pay out-of-pocket in the absence of third-party coverage; and |
• |
physicians or patients may be reluctant to switch from existing therapies even if potentially more effective, safe or convenient. |
Ocuphire has not yet sold any of its products. Ocuphire cannot assure investors that there is a sufficient market demand for its products. Achieving market acceptance for its products will require substantial marketing efforts and expenditure of funds to create awareness and demand by participants in the industry. Ocuphire has not conducted any independent market research to determine the extent of any demand that exists for the products to be provided by it and there is no guarantee that a sufficient interest in the market will exist for the products and services being produced by, or for, it. Any lack of sufficient demand for the products contemplated to be provided by Ocuphire will have a material adverse effect on it.
If the FDA or a comparable foreign regulatory authority approves generic versions of Ocuphire’s product candidates that receive marketing approval, or if such authorities do not grant Ocuphire’s product candidates appropriate periods of exclusivity before approving generic versions of Ocuphire’s products, the sales of Ocuphire’s products could be adversely affected.
Once an NDA is approved, the product covered thereby becomes a “reference listed drug” in the FDA’s publication, “Approved Drug Products with Therapeutic Equivalence Evaluations.” Manufacturers may seek approval of generic versions of reference listed drugs through submission of abbreviated new drug applications (“ANDAs”) in the United States. In support of an ANDA, a generic manufacturer need not conduct clinical studies. Rather, the applicant generally must show that its product has the same active ingredient(s), dosage form, strength, route of administration, and conditions of use or labeling as the reference listed drug (“RLD”) and that the generic version is bioequivalent to the RLD, meaning it is absorbed in the body at the same rate and to the same extent. Generic products may be significantly less costly to bring to market than the RLD, and companies that produce generic products are generally able to offer them at lower prices. Thus, following the introduction of a generic drug, a significant percentage of the sales of any branded product or RLD may be lost to the generic product.
The FDA may not approve an ANDA for a generic product until any applicable period of non-patent exclusivity for the reference listed drug has expired. The FDC Act provides a period of five years of non-patent exclusivity for a new drug containing a new chemical entity (“NCE”). Specifically, in cases where such exclusivity has been granted, an ANDA may not be filed with the FDA until the expiration of five years unless the submission is accompanied by a Paragraph IV certification that a patent covering the reference listed drug is either invalid or will not be infringed by the generic product, in which case the applicant may submit its application four years after approval of the RLD. It is unclear whether the FDA will treat the active ingredients in its product candidates as NCEs
26
and, therefore, afford them five years of NCE exclusivity if they are approved. If any product Ocuphire develops does not receive five years of NCE exclusivity, it may nonetheless be eligible for three years of exclusivity, which means that the FDA may approve generic versions of such product three years after its date of approval. Manufacturers may seek to launch these generic products following the expiration of the applicable marketing exclusivity period, even if Ocuphire still has patent protection for its product.
Competition that Ocuphire’s product candidates would face from generic versions could materially and adversely impact its future revenue, profitability, and cash flows and substantially limit its ability to obtain a return on the investments it has made in any such product candidate.
Even if Ocuphire is able to commercialize its product candidates, their profitability will likely depend in significant part on third-party reimbursement practices, which, if unfavorable, would harm its business.
Ocuphire’s ability to commercialize a drug successfully will depend in part on the extent to which coverage and adequate reimbursement will be available from government health administration authorities, private health insurers and other organizations. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which medications they will pay for and establish reimbursement levels. Government authorities and third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for certain medications. Increasingly, third-party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the prices charged for medical products. Ocuphire cannot be sure that coverage will be available for any product candidate that Ocuphire commercializes and, if coverage is available, whether the level of reimbursement will be adequate. Assuming Ocuphire obtains coverage for its product candidates, if approved, by a third-party payor, the resulting reimbursement payment rates may not be adequate or may require co-payments that patients find unacceptably high. Patients who are prescribed medications for the treatment of their conditions, and their prescribing physicians, generally rely on third-party payors to reimburse all or some of the costs associated with their prescription drugs. Patients are unlikely to use a product candidate, if approved, unless coverage is provided and reimbursement is adequate to cover all or a significant portion of the cost of its products. Therefore, coverage and adequate reimbursement are critical to new product acceptance. If reimbursement is not available or is available only to limited levels, Ocuphire may not be able to successfully commercialize any product candidate for which it obtains marketing approval.
There may be significant delays in obtaining reimbursement for newly approved drugs, and coverage may be more limited than the purposes for which a product candidate is approved by the FDA or similar regulatory authorities outside the United States. Moreover, eligibility for reimbursement does not imply that any product will be paid for in all cases or at a rate that covers Ocuphire’s costs, including research, development, manufacture, sale, and distribution. Interim reimbursement levels for a new product, if applicable, may also not be sufficient to cover Ocuphire’s costs and may not be made permanent. Reimbursement rates may vary according to the use of the product and the clinical setting in which it is used, may be based on reimbursement levels already set for lower cost medicines, and may be incorporated into existing payments for other services. Net prices for products may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and by any future relaxation of laws that presently restrict imports of medicines from countries where they may be sold at lower prices than in the United States. Third-party payors often rely upon Medicare coverage policy and payment limitations in setting their own reimbursement policies. However, there is no uniform policy requirement for coverage and reimbursement for drug products among third-party payors in the United States. Therefore, coverage and reimbursement for drug products can differ significantly from payor to payor. As a result, the coverage determination process is often time-consuming and costly, and it will require Ocuphire to provide scientific and clinical support for the use of its products to each payor separately. There is no assurance that coverage and adequate reimbursement will be applied consistently or obtained in the first instance.
Any inability to promptly obtain coverage and profitable payment rates from government-funded or private payors for any approved products that Ocuphire develops could have an adverse effect on its operating results, its ability to raise capital needed to commercialize products, and its overall financial condition.
Product liability lawsuits against Ocuphire, or its suppliers and manufacturers, could cause it to incur substantial liabilities and could limit commercialization of any product candidate that it may develop.
Ocuphire faces an inherent risk of product liability exposure related to the testing of its product candidates in human clinical trials and will face an even greater risk if it commercially sells any products that it may develop. Product liability claims might be brought against Ocuphire by patients, healthcare providers, or others selling or
27
otherwise coming into contact with its product candidates during product testing, manufacturing, marketing, or sale. For example, Ocuphire may be sued under allegations that a product candidate caused injury or that the product was otherwise unsuitable. Any such product liability claims may include allegations of manufacturing or design defects, failure to warn of dangers inherent in the product, such as interactions with alcohol or other drugs, negligence, or breach of warranty. Claims could also be asserted under state consumer protection acts. If Ocuphire cannot successfully defend itself against claims that its product candidate caused injuries, it could incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
• |
decreased demand for any product candidate that Ocuphire is developing; |
• |
injury to Ocuphire’s reputation and significant negative media attention; |
• |
withdrawal of clinical trial participants; |
• |
increased FDA warnings on product labels; |
• |
significant costs to defend the related litigation; |
• |
substantial monetary awards to trial participants or patients; |
• |
distraction of management’s attention from Ocuphire’s primary business; |
• |
loss of revenue; and |
• |
the inability to commercialize any product candidate that Ocuphire may develop. |
Its product liability and/or clinical trial insurance coverage may not be adequate to cover all liabilities that Ocuphire may incur. Ocuphire may need to increase its insurance coverage as it expands clinical trials and if it successfully commercializes its product candidates. Insurance coverage is increasingly expensive, and it may not be able to obtain product liability insurance on commercially reasonable terms or for a sufficient amount to satisfy liabilities that may arise.
Similarly, Ocuphire may be a party to, or may be otherwise responsible for, pending or threatened lawsuits or other claims related to products purchased from its manufacturers and suppliers. Although Ocuphire intends to require its providers to have product liability insurance, the ability to obtain such coverage and the sufficiency thereof is uncertain. Such cases and claims may raise difficult and complex factual and legal issues and may be subject to many uncertainties and complexities, including, but not limited to, the facts and circumstances of each particular case or claim, the jurisdiction in which each suit is brought, and differences in applicable law. Such litigation could result in additional expense and exposure in excess of Ocuphire’s anticipated reserves, especially if such matters are not covered by insurance. Upon resolution of any pending legal matters or other claims, Ocuphire may incur charges in excess of established reserves. Product liability lawsuits and claims, safety alerts or product recalls in the future, regardless of their ultimate outcome, could have a material adverse effect on the business and reputation and on Ocuphire’s ability to attract and retain customers and strategic partners. The business, profitability and growth prospects could suffer if Ocuphire faces such negative publicity.
If Ocuphire or its third-party manufacturers fail to comply with environmental or health and safety laws and regulations, Ocuphire could become subject to fines or penalties or incur costs that could have an adverse effect on the success of its business.
Ocuphire’s research and development activities involve the controlled use of potentially hazardous substances, including chemical and biological materials, by itself and its third-party manufacturers. Ocuphire’s manufacturers are subject to federal, state, and local laws and regulations in the United States and abroad governing laboratory procedures and the use, manufacture, storage, handling, and disposal of medical and hazardous materials. Although Ocuphire believes that its manufacturers’ procedures for using, handling, storing, and disposing of these materials comply with legally prescribed standards, it cannot eliminate the risk of contamination or injury resulting from medical or hazardous materials. As a result of any such contamination or injury, Ocuphire may incur liability, or federal, state, city, or local authorities may curtail its use of these materials and interrupt its business operations. In the event of an accident, Ocuphire could be held liable for damages or fined, and such liability or fines could exceed its resources. Ocuphire does not have insurance for liabilities arising from medical or hazardous materials. Although Ocuphire maintains workers’ compensation insurance for costs and expenses that it may incur due to injuries to its employees resulting from the use of hazardous materials, this insurance may not provide adequate coverage against potential liabilities. Compliance with applicable environmental and health and safety laws and regulations is
28
expensive, and current or future environmental regulations may impair Ocuphire’s research, development, and production efforts, which could harm its business, prospects, financial condition, or results of operations.
Federal legislation and actions by state and local governments could permit reimportation of drugs from foreign countries into the United States, which could adversely affect Ocuphire’s operating results when the drugs are sold at lower prices in foreign countries than in the United States.
Ocuphire may face competition for its product candidates, if approved, from other therapies sourced from foreign countries that have price controls on pharmaceutical products. The Medicare Modernization Act contains provisions that may change U.S. reimportation laws and expand pharmacists’ and wholesalers’ ability to import cheaper versions of approved drugs or competing products from Canada, where there are government price controls. These changes to U.S. importation laws would not take effect unless and until the Secretary of Health and Human Services certifies that the changes would pose no additional risk to the public’s health and safety and would result in a significant reduction in the cost of products to consumers. The Secretary of Health and Human Services has so far declined to approve a reimportation plan. Proponents of drug reimportation may attempt to pass legislation that would directly allow reimportation under certain circumstances. Legislation or regulations allowing the reimportation of drugs, if enacted, could decrease the price Ocuphire receives for any product it may develop and adversely affect its future revenues and prospects for profitability.
Risks Related to Ocuphire’s Reliance on Third Parties
Ocuphire will be unable to directly control all aspects of its clinical trials due to its reliance on clinical research organizations (CROs) and other third parties that assist Ocuphire in conducting clinical trials.
Ocuphire relies on third party CROs and other third parties to assist in managing, monitoring, and otherwise carrying out its clinical trials. Ocuphire expects to continue to rely on third parties, such as CROs, clinical data management organizations, medical institutions, and clinical investigators, to conduct its clinical trials in the future, including its Phase 3 development program for Nyxol. Ocuphire competes with many other companies for the resources of these third parties.
As a result, Ocuphire will have limited control over the conduct, timing, and completion of these clinical trials and the management of data developed through the clinical trials. Communicating with outside parties can also be challenging, potentially leading to mistakes as well as difficulties in coordinating activities. Additionally, the ongoing COVID-19 pandemic may affect the ability of third parties to fulfill their obligations to Ocuphire. Outside parties may:
• |
have staffing difficulties; |
• |
fail to comply with contractual obligations; |
• |
experience regulatory compliance issues; |
• |
undergo changes in priorities or become financially distressed; or |
• |
form relationships with other entities, some of which may be Ocuphire’s competitors. |
These factors may adversely affect the willingness or ability of third parties to conduct Ocuphire’s clinical trials and may subject Ocuphire to unexpected cost increases that are beyond its control.
While Ocuphire’s reliance on these third parties for research and development activities will reduce its control over these activities, it will not relieve Ocuphire of its responsibilities and requirements. For example, the FDA requires Ocuphire to comply with standards, commonly referred to as good clinical practices (“GCP”), for conducting, recording, and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity, and confidentiality of clinical trial participants are protected.
Problems with the timeliness or quality of the work of any CRO may lead Ocuphire to seek to terminate its relationship with any such CRO and use an alternative service provider. Making this change may be costly or delay Ocuphire’s clinical trials, and contractual restrictions may make such a change difficult or impossible. If Ocuphire must replace any CRO that is conducting its clinical trials, its clinical trials may have to be suspended until it finds another CRO that offers comparable services. The time that it would take Ocuphire to find alternative organizations may cause a delay in the commercialization of its product candidates, or it may cause it to incur significant expenses to replicate any lost data. Although Ocuphire does not believe that any CRO on which it would rely would offer
29
services that are not available elsewhere, it may be difficult to find a replacement organization that can conduct Ocuphire’s clinical trials in an acceptable manner and at an acceptable cost. Any delay in or inability to complete Ocuphire’s clinical trials could significantly compromise its ability to secure regulatory approval for its product candidates and preclude its ability to commercialize its product candidates, thereby limiting or preventing its ability to generate sales revenue.
Ocuphire relies completely on third parties to supply and manufacture its preclinical and clinical drug supplies for product candidates, and intends to rely on third parties to produce commercial supplies of its current and any future product candidates.
Ocuphire does not currently have, nor does it plan to acquire, the infrastructure or capability to internally manufacture its clinical drug supply of product candidates for use in the conduct of its preclinical studies and clinical trials. Ocuphire lacks the internal resources and the capability to manufacture any product candidates on a clinical or commercial scale. The process of manufacturing drug products is complex, highly regulated, and subject to several risks. For example, the facilities used by Ocuphire’s contract manufacturers to manufacture the active pharmaceutical ingredient (or drug substance) and final drug product for product candidates must be inspected by the FDA and other comparable foreign regulatory agencies in connection with Ocuphire’s submission of an NDA or relevant foreign regulatory submission to the applicable regulatory agency. In addition, the manufacturing of drug substance or product is susceptible to product loss due to contamination, equipment failure, improper installation or operation of equipment, or vendor or operator error. Moreover, the manufacturing facilities in which product candidates are made could be adversely affected by equipment failures, labor shortages, natural disasters, power failures, or other factors.
Ocuphire does not control the manufacturing process of its contract manufacturers, and is completely dependent on them to comply with current good manufacturing practices (“cGMP”) for manufacture of both active drug substances and finished drug products. If Ocuphire’s contract manufacturers cannot successfully manufacture material that conforms to its specifications and the strict regulatory requirements of the FDA or applicable foreign regulatory agencies, Ocuphire will not be able to secure and/or maintain regulatory approval for its products. In addition, Ocuphire has no direct control over its contract manufacturers’ ability to maintain adequate quality control, quality assurance, and qualified personnel. Failure to satisfy the regulatory requirements for the production of those materials and products may affect the regulatory clearance of Ocuphire’s contract manufacturers’ facilities generally. If the FDA or a comparable foreign regulatory agency does not approve these facilities for the manufacture of product candidates, or if it withdraws its approval in the future, Ocuphire may need to find alternative manufacturing facilities, which would adversely impact Ocuphire’s ability to develop, obtain regulatory approval for, or market product candidates. Furthermore, all of Ocuphire’s contract manufacturers are engaged with other companies to supply and/or manufacture materials or products for such companies, which exposes its manufacturers to regulatory and sourcing risks for the production of such materials and products. To the extent practicable, Ocuphire attempts to identify more than one supplier. However, some raw materials are available only from a single source or only one supplier has been identified, even in instances where multiple sources exist.
Ocuphire has relied and will rely upon third-party manufacturers in the United States and overseas for the manufacture of Nyxol and APX3330 for preclinical and clinical testing purposes and intends to continue to do so in the future for Nyxol, APX3330, the combination kit of Nyxol and low-dose pilocarpine, and any other product candidates, including for commercial purposes. If Ocuphire’s third-party manufacturers are unable to supply drug substance and/or drug product on a commercial basis, Ocuphire may not be able to successfully produce and market product candidates, if approved, or it could be delayed in doing so. For instance, Ocuphire presently relies on one supplier in Italy for the drug substance for Nyxol, and one manufacturer in India for APX3330 drug substance. If there is any delay or problem with the manufacture of these drug substance or if there is a delay in producing finished drug product from these drug substances, the development and possible approval of Ocuphire’s product candidates and potential commercial launch may be delayed or otherwise adversely affected. Ocuphire will rely on comparison of product specifications (identity, strength, quality, and potency) to demonstrate equivalence of the current drug substance and/or drug product to the drug substance and/or drug product used in previously completed preclinical and clinical testing. If Ocuphire is unable to demonstrate such equivalence, it may be required to conduct additional preclinical and/or clinical testing of its product candidates. The formulation of the low-dose pilocarpine in the combination product candidate of Nyxol is still in development. Also, due to the current COVID-19 pandemic, disruptions of global supply chains are more likely to occur, which could delay the clinical development of Ocuphire’s product candidates. Ocuphire has already experienced a few interruptions in its manufacturing, supply chain, research and development operations, regulatory and financial position, including, for example, the acceleration of the shipment of active pharmaceutical ingredient supply from overseas.
30
Due to these and other potential problems, Ocuphire is exploring the possibility of establishing additional sources of supply, with U.S. manufacturers, for the active pharmaceutical ingredients of both Nyxol and APX3330. Establishing these additional sources, including qualifying their manufacturing processes and demonstrating the equivalence of their products, may be costly, time-consuming, and difficult to effectuate, and may delay Ocuphire’s research and development activities. If Ocuphire must replace any manufacturer, its research and development activities may have to be suspended until it finds another manufacturer that offers comparable services. The time that it takes Ocuphire to find alternative organizations may cause a delay in the development and commercialization of product candidates.
Ocuphire may form or seek strategic alliances or enter into licensing arrangements in the future, and may not realize benefits from such alliances or licensing arrangements.
Ocuphire may form or seek strategic alliances, create joint ventures or collaborations or enter into additional licensing arrangements with third parties that it believes will complement or augment its development and commercialization efforts with respect to product candidates. Any of these relationships may require Ocuphire to incur non-recurring and other charges, increase its near- and long-term expenditures, or issue securities that dilute Ocuphire’s existing stockholders, which may disrupt its management and business. Ocuphire’s likely collaborators include large, mid-size, regional, or national pharmaceutical companies and biotechnology companies. If Ocuphire enters into any such arrangements with any third parties, it will likely have limited control over the amount and timing of resources that its collaborators dedicate to the development or commercialization of product candidates. Ocuphire’s ability to generate revenues from these arrangements will depend on its collaborators’ abilities to successfully perform the functions assigned to them in these arrangements. Ocuphire cannot be certain that, following a strategic transaction or license, it will achieve the revenue or specific net income that justifies such transaction. Collaborations involving product candidates pose the following risks to Ocuphire:
• |
collaborators have significant discretion in determining the efforts and resources that they will apply to these collaborations; |
• |
collaborators may not perform their obligations as expected; |
• |
collaborators may not pursue development and commercialization or may elect not to continue or renew development or commercialization programs based on clinical trial results, changes in the collaborator’s strategic focus or available funding, or external factors such as an acquisition that diverts resources or creates competing priorities; |
• |
collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials, or require a new formulation of a product candidate for clinical testing; |
• |
collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with its product candidate if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more attractive than Ocuphire’s; |
• |
a collaborator with marketing and distribution rights to one or more product candidates may not commit sufficient resources to the marketing or distribution of any such product candidate; |
• |
collaborators may not properly maintain or defend Ocuphire’s intellectual property rights or may use its proprietary information in such a way as to invite litigation that could jeopardize or invalidate Ocuphire’s proprietary information or expose Ocuphire to litigation; |
• |
collaborators may infringe the intellectual property rights of third parties, which may expose Ocuphire to litigation and potential liability; |
• |
disputes may arise between the Ocuphire and collaborators that result in the delay or termination of research, development, or commercialization of its product candidates, or in litigation or arbitration that diverts management attention and resources; |
• |
Ocuphire may lose certain valuable rights under circumstances identified in its collaborations, including if it undergoes a change of control; |
31
• |
collaborations may be terminated and such terminations may create a need for additional capital to pursue further development or commercialization of the applicable product candidates; |
• |
collaborators may learn about Ocuphire’s discoveries and use this knowledge to compete with Ocuphire in the future; |
• |
the results of collaborators’ preclinical or clinical studies could harm or impair other development programs; |
• |
there may be conflicts between different collaborators that could negatively affect those collaborations and potentially others; |
• |
the number and nature of Ocuphire’s collaborations could adversely affect its attractiveness to potential future collaborators or acquirers; |
• |
collaboration agreements may not lead to development or commercialization of its product candidate in the most efficient manner or at all. If a present or future collaborator of Ocuphire were to be involved in a business combination, the continued pursuit and emphasis on its product development or commercialization program under such collaboration could be delayed, diminished, or terminated; and |
• |
collaborators may be unable to obtain the necessary marketing approvals. |
If future collaboration partners fail to develop or effectively commercialize product candidates for any of these reasons, such product candidates may not be approved for sale and Ocuphire’s sales of such product candidates, if approved, may be limited, which would have an adverse effect on Ocuphire’s operating results and financial condition.
If Ocuphire is not able to establish new collaborations on commercially reasonable terms, it may have to alter its development, manufacturing, and commercialization plans.
Ocuphire faces significant competition in attracting collaborators for development, manufacturing or commercialization plans. Whether it reaches a definitive agreement for collaboration will depend, among other things, upon its assessment of the proposed collaborator’s resources, expertise, and evaluation of a number of factors related to the associated product candidate, as well as the terms and conditions of the proposed collaboration. Those factors may include the design or results of clinical trials, the likelihood of approval by the FDA or similar regulatory authorities outside the United States, the potential market for the product candidate, the costs and complexities of manufacturing and delivering such product candidate to patients, the potential of competing products, the existence of uncertainty with respect to Ocuphire’s ownership of technology, which may exist if there is a challenge to such ownership without regard to the merits of the challenge, and industry and market conditions generally. The collaborator may also consider alternative product candidates or technologies for similar indications that may be available for collaborations and whether such a collaboration could be more attractive than one with Ocuphire. Ocuphire may not be able to enter into these agreements on commercially reasonable terms, or at all.
Much of the potential revenue from future commercial collaborations may consist of contingent payments, such as payments for achieving regulatory milestones or royalties payable on sales of Ocuphire’s product candidate, if approved. The milestone and royalty revenue that Ocuphire may receive under these collaborations would depend upon its collaborators’ ability to successfully develop, introduce, market and sell its product candidate, if approved. In addition, collaborators may decide to enter into arrangements with third parties to commercialize products developed under collaborations related to its product candidate, which could reduce the milestone and royalty revenue received, if any.
Ocuphire may also be restricted under existing collaboration agreements from entering into future agreements on certain terms with potential collaborators. Collaborations are complex and time-consuming to negotiate and document. In addition, there have been a significant number of recent business combinations among large pharmaceutical companies that have resulted in a reduced number of potential future collaborators.
Ocuphire may not be able to negotiate collaborations on a timely basis and on acceptable terms, or at all. If Ocuphire is unable to do so, it may have to curtail the development of the product candidate for which it is seeking to collaborate, reduce or delay its development program or that of one or more of its other development programs, delay its potential commercialization or reduce the scope of any sales or marketing activities, or increase its expenditures and undertake development or commercialization activities at its own expense. If Ocuphire elects to increase its expenditures to fund development or commercialization activities on its own, it may need to obtain
32
additional capital, which may not be available to Ocuphire on acceptable terms or at all. If Ocuphire does not have sufficient funds, it may not be able to further develop its product candidate or bring it to market and generate product revenue.
If Ocuphire engages in acquisitions, in-licensing or strategic partnerships, this may increase its capital requirements, dilute its stockholders, cause it to incur debt or assume contingent liabilities and subject it to other risks.
Ocuphire may engage in various acquisitions and strategic partnerships, including licensing or acquiring complementary products, intellectual property rights, technologies, or businesses. Any acquisition or strategic partnership may entail numerous risks, including:
• |
increased operating expenses and cash requirements; |
• |
the assumption of indebtedness or contingent liabilities; |
• |
the issuance of Ocuphire’s equity securities which would result in dilution to Ocuphire Stockholders; |
• |
assimilation of operations, intellectual property, products and product candidates of an acquired company, including difficulties associated with integrating new personnel; |
• |
the diversion of management’s attention from Ocuphire’s existing product candidates and initiatives in pursuing such an acquisition or strategic partnership; |
• |
retention of key employees, the loss of key personnel, and uncertainties in Ocuphire’s ability to maintain key business relationships; |
• |
risks and uncertainties associated with the other party to such a transaction, including the prospects of that party and their existing products or product candidates and regulatory approvals; and |
• |
Ocuphire’s inability to generate revenue from acquired intellectual property, technology and/or products sufficient to meet its objectives or even to offset the associated transaction and maintenance costs. |
In addition, if Ocuphire undertakes such a transaction, it may incur large one-time expenses and acquire intangible assets that could result in significant future amortization expense.
Risks Related to Ocuphire’s Intellectual Property
If Ocuphire is unable to obtain and maintain sufficient patent protection for its product candidates, its competitors could develop and commercialize products or technology similar or identical to those of Ocuphire, which would adversely affect Ocuphire’s ability to successfully commercialize any product candidates it may develop, its business, results of operations, financial condition and prospects.
Ocuphire primarily protects its intellectual property through a combination of patents and patent applications on inventions, trademark protection on its product name, and trade secret protection as it deems appropriate.
The patent estate relating to Ocuphire’s Nyxol product candidate contains ten U.S. patents, four pending U.S. non-provisional patent applications, two pending international patent applications, as well as issued patents in Australia, Europe, Japan, and Mexico, and pending patent applications in Canada, all of which are owned by Ocuphire.
Ocuphire’s U.S. Patents 9,795,560, 10,278,918 and 10,772,829 and counterpart Australian, European, and Japanese patents each contain composition of matter claims to aqueous phentolamine mesylate formulations and are scheduled to expire in year 2034. A counterpart patent application directed to aqueous phentolamine mesylate formulations is pending in Canada, where a patent, if granted, based on this pending patent application, would expire in year 2034. In the same patent family, there is one pending U.S patent application with additional claims to aqueous phentolamine mesylate formulations, whereby a patent, if granted based on this patent application, would expire in year 2034. The patents and patent applications cover the current clinical formulation for the Nyxol product.
Ocuphire’s U.S. Patent Nos. 9,089,560 and 9,789,088 contain claims directed to methods of improving visual performance using, for example, phentolamine mesylate and are scheduled to expire in year 2034. Counterpart patents have issued in Australia, Europe and Japan, which are scheduled to expire in year 2034. Counterpart patent applications are pending in Canada, along with a further patent application pending in the U.S. Patents, if granted from these pending patent applications, would expire in year 2034. The patents and patent applications cover uses of the current clinical formulation for the Nyxol product.
33
Ocuphire’s pending international patent application PCT/US2019/056324 is directed to treating glaucoma and other medical disorders using phentolamine mesylate. Patents, if granted based on this pending patent application, would expire in year 2039. Ocuphire’s pending international patent application PCT/US2019/058182 is directed to methods of treating presbyopia, mydriasis, and other medical disorders; patents, if granted based on this pending patent application, would expire in year 2039. Two pending U.S. patent applications have been filed based on international patent application PCT/US2019/058182, one with claims to treating presbyopia and the other with claims to treating mydriasis.
The remaining five of Ocuphire’s U.S. patents are scheduled to expire in year 2020 and have claims to methods of use or ophthalmic formulations containing an ophthalmic artificial tear solution, which is not the current clinical formulation used in the Nyxol product. Ocuphire’s issued patent in Mexico is scheduled to expire in year 2025 and has claims to ophthalmic formulations.
Ocuphire has in-licensed a patent estate directed to APX3330 and related compounds that contains five U.S. patents, four pending U.S. non-provisional patent applications, and one pending international patent application, as well as issued patents in Europe, Japan, Canada, and Australia, and pending patent applications in Europe, Japan, China, and Canada. Ocuphire’s in-licensed U.S. patent 9,040,505 has claims to methods of treating diabetic retinopathy and other diseases using, for example, APX3330 and is scheduled to expire in year 2030. Counterpart patents have issued in Europe, Japan, Australia, and Canada, which are scheduled to expire in year 2028, and there is a related pending U.S. patent application with method of treatment claims that, if issued as a patent, would expire in year 2028. Ocuphire’s in-licensed pending international patent application PCT/US2019/017023 has claims to methods of treating wAMD and other diseases using, for example, APX3330. Patents, if granted based on this pending international patent application, would expire in year 2039. Ocuphire’s in-licensed patent applications directed to a combination therapy composition comprising an APE1/REF-1 inhibitor, such as APX3330, and a second therapeutic agent, and methods of using such combination therapies to treat retinal diseases and other indications are pending in the U.S., Europe, Japan, China, and Canada, whereby patents, if granted based on these pending patent applications, would expire in year 2038. Patents to derivatives of APX3330 have issued in the U.S., Europe, and other countries that are scheduled to expire from year 2028 to 2032, and patent applications to derivatives of APX3330 are pending in the U.S., Europe, and other countries whereby a patent, if granted based on these pending patent applications, would expire from year 2028 to 2032.
The patent prosecution process is expensive and time-consuming, and Ocuphire and its future licensors, licensees, or collaboration partners may not be able to prepare, file, and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that Ocuphire or any future licensors, licensees, or collaboration partners may fail to identify patentable aspects of inventions made in the course of development and commercialization activities before it is too late to obtain patent protection on them. Ocuphire and its licensors’ patent applications cannot be enforced against third parties practicing the technology claimed in such applications unless and until a patent issues from such applications, and then only to the extent the issued claims cover the technology.
Ocuphire cannot assure you that any of its patents have matured, or that any of its pending patent applications will mature, into issued patents that will include, claims with a scope sufficient to protect its product candidates. Others have developed technologies that may be related or competitive to Ocuphire’s approach, and may have filed or may file patent applications and may have received or may receive patents that overlap or conflict with Ocuphire’s patent applications, for example by claiming the same compounds, methods or formulations or by claiming subject matter that could dominate the patents that Ocuphire owns or in-licenses. The patent positions of biotechnology and pharmaceutical companies, including Ocuphire’s patent position, involve complex legal and factual questions, and, therefore, the issuance, scope, validity, and enforceability of any patent claims that Ocuphire may obtain cannot be predicted with certainty. Patents, if issued, may be challenged, deemed unenforceable, invalidated, or circumvented. U.S. patents and patent applications may also be subject to interference proceedings, ex parte reexamination, or inter partes review proceedings, supplemental examination and challenges in district court. Patents may be subjected to opposition, post-grant review, or comparable proceedings in various national and regional patent offices. These proceedings could result in either loss of the patent or denial of the patent application or loss or reduction in the scope of one or more of the claims of the patent or patent application. In addition, such interference, re-examination, opposition, post-grant review, inter partes review, supplemental examination, or revocation proceedings may be
34
costly or time-consuming. Thus, any patents that Ocuphire may own or exclusively license may not provide any protection against competitors. Furthermore, an adverse decision in an interference proceeding can result in a third party receiving the patent right sought by Ocuphire, which in turn could affect its ability to develop, market or otherwise commercialize its product candidates.
Furthermore, the issuance of a patent, while presumed valid, is not conclusive as to its validity or its enforceability and it may not provide Ocuphire with adequate proprietary protection or competitive advantages against competitors with similar products. Competitors may also be able to design around Ocuphire’s patents. Other parties may develop and obtain patent protection for more effective technologies, designs, or methods. Ocuphire may not be able to prevent the unauthorized disclosure or use of any technical knowledge or trade secrets by consultants, vendors, former employees, or current employees. The laws of some foreign countries do not protect proprietary rights to the same extent as do the laws of the United States, and Ocuphire may encounter significant problems in protecting its proprietary rights in these countries. If these developments were to occur, they could have a material adverse effect on Ocuphire’s sales.
Ocuphire’s ability to enforce its patent rights depends on its ability to detect infringement. It is difficult to detect infringers who do not advertise the components that are used in their products. Moreover, it may be difficult or impossible to obtain evidence of infringement in a competitor’s or potential competitor’s product. Any litigation to enforce or defend Ocuphire’s patent rights, if any, even if Ocuphire were to prevail, could be costly and time-consuming and would divert the attention of management and key personnel from Ocuphire’s business operations. Ocuphire may not prevail in any lawsuits that it initiates and the damages or other remedies awarded if it were to prevail may not be commercially meaningful.
In addition, proceedings to enforce or defend Ocuphire’s patents could put its patents at risk of being invalidated, held unenforceable, or interpreted narrowly. Such proceedings could also provoke third parties to assert claims against Ocuphire, including that some or all of the claims in one or more of Ocuphire’s patents are invalid or otherwise unenforceable. If, in any proceeding, a court invalidated or found unenforceable Ocuphire’s patents covering its product candidates, Ocuphire’s financial position and results of operations would be adversely impacted. In addition, if a court found that valid, enforceable patents held by third parties covered Ocuphire’s product candidates, its financial position and results of operations would also be adversely impacted.
The degree of future protection for Ocuphire’s proprietary rights is uncertain, and Ocuphire cannot ensure that:
• |
any of Ocuphire’s patents, or any of its pending patent applications, if issued, will include claims having a scope sufficient to protect its product candidates; |
• |
any of its pending patent applications will result in issued patents; |
• |
Ocuphire will be able to successfully commercialize its product candidates, if approved, before its relevant patents expire; |
• |
Ocuphire was the first to make the inventions covered by each of its patents and pending patent applications; |
• |
Ocuphire was the first to file patent applications for these inventions; |
• |
others will not develop similar or alternative technologies that do not infringe Ocuphire’s patents; |
• |
any of Ocuphire’s patents will be valid and enforceable; |
• |
any patents issued to Ocuphire will provide a basis for an exclusive market for its commercially viable products, will provide Ocuphire with any competitive advantages or will not be challenged by third parties; |
• |
Ocuphire will develop additional proprietary technologies or product candidates that are separately patentable; or |
• |
that Ocuphire’s commercial activities or products will not infringe upon the patents of others. |
Patents have a limited lifespan. The natural expiration of a patent is generally 20 years after its effective filing date. Various extensions may be available; however, the life of a patent, and the protection it affords, is limited. Given the extensive period of time between patent filing and regulatory approval for a product candidate, the time during
35
which Ocuphire can market a product candidate under patent protection is limited, and Ocuphire’s patent may expire before it obtains such approval. Without patent protection for its product candidates, it may be vulnerable to competition from generic versions of its product candidates, which may affect the profitability of its product candidates.
If Ocuphire does not obtain protection under the Hatch-Waxman Act and similar foreign legislation by extending the patent terms and obtaining data exclusivity for its product candidate, its business may be materially harmed.
Depending upon the timing, duration of regulatory review, and date of FDA marketing approval of its APX3330 or other product candidates, if any, one of such U.S. patents may be eligible for patent term restoration under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch-Waxman Act. The Hatch-Waxman Act provides for a patent restoration term, or patent term extension, of up to five years as compensation for the time the product is under FDA regulatory review. The duration of patent term extension is calculated based on the time spent in the regulatory review process. In the future, Ocuphire may plan to seek patent term extension for one or more of its patents related to its APX3330 or other product candidates. However, Ocuphire may not be granted an extension because of, for example, failing to apply within the applicable deadline, expiration of relevant patents prior to obtaining approval, or otherwise failing to satisfy applicable requirements. Moreover, the applicable time period or the scope of patent protection afforded could be shorter or less than what Ocuphire requests. If Ocuphire is unable to obtain patent term extension or the term of any such extension is less than it requests, Ocuphire’s revenue could be reduced, possibly materially.
Changes in U.S. patent law could diminish the value of patents in general, thereby impairing Ocuphire’s ability to protect its product candidates.
In 2011, the United States enacted wide-ranging patent reform legislation with the America Invents Act (“AIA”).
An important change introduced by the AIA is that, as of March 16, 2013, the United States transitioned to a “first-to-file” system for deciding which party should be granted a patent when two or more patent applications are filed by different parties claiming the same invention. A third party that files a patent application in the USPTO after that date but before Ocuphire could therefore be awarded a patent covering an invention of ours even if Ocuphire had made the invention before it was made by the third party. This will require Ocuphire to be cognizant going forward of the time from invention to filing of a patent application, but circumstances could prevent Ocuphire from promptly filing patent applications on its inventions.
Among some of the other changes introduced by the AIA are changes that limit where a patentee may file a patent infringement suit and providing opportunities for third parties to challenge any issued patent in the USPTO. This applies to all of Ocuphire’s U.S. patents, even those issued before March 16, 2013. Because of a lower evidentiary standard in USPTO proceedings compared to the evidentiary standard in U.S. federal courts necessary to invalidate a patent claim, a third party could potentially provide evidence in a USPTO proceeding sufficient for the USPTO to hold a claim invalid even though the same evidence would be insufficient to invalidate the claim if first presented in a district court action. Accordingly, a third party may attempt to use the USPTO procedures to invalidate Ocuphire’s patent claims that would not have been invalidated if first challenged by the third party as a defendant in a district court action. The AIA and its implementation could increase the uncertainties and costs surrounding the prosecution of Ocuphire’s patent applications and the enforcement or defense of Ocuphire’s issued patents.
Additionally, the U.S. Supreme Court’s holdings in several patent cases in recent years, such as Association for Molecular Pathology v. Myriad Genetics, Inc. (Myriad I), Mayo Collaborative Services v. Prometheus Laboratories, Inc., and Alice Corporation Pty. Ltd. v. CLS Bank International, have narrowed the scope of patent protection available in certain circumstances or weakened the rights of patent owners in certain situations. In addition to increasing uncertainty about to Ocuphire’s ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by the U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways that would weaken Ocuphire’s ability to obtain new patents or to enforce Ocuphire’s existing patents and patents that it might obtain in the future.
Ocuphire may not be able to protect or practice its intellectual property rights throughout the world.
In jurisdictions where Ocuphire has not obtained patent protection, competitors may use its intellectual property to develop their own products and further, may export otherwise infringing products to territories where Ocuphire has
36
patent protection, but where it is more difficult to enforce a patent as compared to the United States. Competitor products may compete with Ocuphire’s product candidates in jurisdictions where it does not have issued or granted patents or where its issued or granted patent claims or other intellectual property rights are not sufficient to prevent competitor activities in these jurisdictions. The legal systems of certain countries, particularly certain developing countries, make it difficult to enforce patents and such countries may not recognize other types of intellectual property protection, particularly that relating to pharmaceuticals. This could make it difficult for Ocuphire to prevent the infringement of its patents or marketing of competing products in violation of its proprietary rights generally in certain jurisdictions. Proceedings to enforce Ocuphire’s patent rights in foreign jurisdictions could result in substantial cost and divert its efforts and attention from other aspects of its business.
The laws of some jurisdictions do not protect intellectual property rights to the same extent as the laws in the United States, and many companies have encountered significant difficulties in protecting and defending such rights in such jurisdictions. If Ocuphire, or any future licensor, encounters difficulties in protecting, or is otherwise precluded from effectively protecting, the intellectual property rights important for its business in such jurisdictions, the value of these rights may be diminished and Ocuphire may face additional competition from others in those jurisdictions. Many countries have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties. In addition, many countries limit the enforceability of patents against government agencies or government contractors. In these countries, the patent owner may have limited remedies, which could materially diminish the value of such patent. If Ocuphire, or any licensor, is forced to grant a license to third parties with respect to any patents relevant to its business, Ocuphire’s competitive position in the relevant jurisdiction may be impaired and its business and results of operations may be adversely affected.
Ocuphire may become involved in lawsuits to protect or enforce its patents and other intellectual property rights, which could be expensive, time consuming, and unsuccessful.
Competitors may infringe Ocuphire’s patents, the patents of its licensing partners, or other intellectual property rights. To counter infringement or unauthorized use, Ocuphire may be required to file infringement claims, which can be expensive and time consuming. In addition, in an infringement proceeding, a court may decide that an Ocuphire patent is invalid or unenforceable, or may refuse to stop the other party from using the technology on the grounds that Ocuphire’s patents do not cover the technology in question. An adverse result in any litigation proceeding could put one or more of Ocuphire’s patents at risk of being invalidated or interpreted narrowly. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of Ocuphire’s confidential information could be compromised by disclosure during this type of litigation. Moreover, there can be no assurance that Ocuphire will have sufficient financial or other resources to file and pursue such infringement claims, which typically last for years before they are concluded.
Litigation proceedings may fail and, even if successful, may be costly and a distraction to Ocuphire’s management and other employees. Ocuphire may not be able to prevent, alone or with its collaborators, misappropriation of its proprietary rights, particularly in countries where the laws may not protect those rights as fully as in the United States.
In addition, there could be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a substantial adverse effect on the price of Ocuphire common stock.
Third parties may initiate legal proceedings alleging that Ocuphire is infringing their intellectual property rights, the outcome of which would be uncertain and could have an adverse effect on the success of Ocuphire’s business.
Ocuphire’s commercial success depends upon its ability and the ability of its collaborators to develop, manufacture, market and sell its product candidates and use its proprietary technologies without infringing the proprietary rights and intellectual property of third parties. The biotechnology and pharmaceutical industries are characterized by extensive litigation regarding patents and other intellectual property rights. Ocuphire may in the future become party to, or threatened with, adversarial proceedings or litigation regarding intellectual property rights with respect to its medicines and technology, including interference or derivation proceedings, post-grant reviews, inter partes reviews, or other procedures before the USPTO or other similar procedures in foreign jurisdictions. Third parties may assert infringement claims against Ocuphire based on existing patents or patents that may be granted in the future. If Ocuphire is found to infringe a third party’s intellectual property rights, it could be required to obtain a license from such third party to continue developing and marketing its medicines and technology. However,
37
Ocuphire may not be able to obtain any required license on commercially reasonable terms or at all. Even if Ocuphire were able to obtain a license, it could be non-exclusive, thereby giving its competitors and other third parties access to the same technologies licensed to it. Ocuphire could be forced, including by court order, to cease developing and commercializing the infringing technology or medicine. In addition, Ocuphire could be held liable for substantial monetary damages, potentially including treble damages and attorneys’ fees, if found to have willfully infringed. A finding of infringement could prevent Ocuphire from commercializing a product candidate or force it to cease some of its business operations, which could harm Ocuphire’s business. Alternatively, Ocuphire may need to redesign its infringing products, which may be impossible or require substantial time and monetary expenditure. Claims that Ocuphire has misappropriated the confidential information or trade secrets of third parties could have a similar negative impact on its business.
The cost to Ocuphire of any litigation or other proceeding relating to patent or other proprietary rights, even if resolved in its favor, could be substantial and may result in substantial costs and distraction to Ocuphire’s management and other employees. Some of Ocuphire’s competitors may be able to sustain the costs of complex patent litigation more effectively than Ocuphire can because they have substantially greater resources. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could delay Ocuphire’s research and development efforts and limit its ability to continue its operations.
Ocuphire may be subject to damages resulting from claims that its employees or Ocuphire has wrongfully used or disclosed alleged trade secrets of their former employers.
Ocuphire’s employees and consultants have been previously employed at other biotechnology or pharmaceutical companies, including its competitors or potential competitors. Although Ocuphire is not aware of any claims currently pending against it, Ocuphire may be subject to claims that these employees or Ocuphire has inadvertently or otherwise used or disclosed trade secrets or other proprietary information or intellectual property of the former employers of its employees. Litigation may be necessary to defend against these claims. Even if Ocuphire is successful in defending against these claims, litigation could result in substantial costs and be a distraction to management. If Ocuphire fails in defending such claims, in addition to paying money claims, it may lose valuable intellectual property rights or personnel. A loss of key personnel or their work product could detract from Ocuphire’s ability to develop or commercialize its product candidates.
If Ocuphire is not able to adequately prevent disclosure of trade secrets and other proprietary information, the value of any product it may pursue could be significantly diminished.
Ocuphire may rely upon trade secrets, know-how, and continuing technological innovation to develop and maintain its competitive position. However, trade secrets are difficult to protect. Ocuphire relies in part on confidentiality agreements with its employees, consultants, outside scientific collaborators, sponsored researchers, contract manufacturers, vendors, and other advisors to protect its trade secrets and other proprietary information. These agreements may not effectively prevent disclosure of confidential information and may not provide an adequate remedy in the event of unauthorized disclosure of confidential information. In addition, Ocuphire cannot guarantee that it has executed these agreements with each party that may have or has had access to trade secrets.
If a party breaches an agreement and discloses Ocuphire’s proprietary information, including its trade secrets, Ocuphire may not be able to obtain adequate remedies for such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive, and time consuming, and the outcome is unpredictable. In addition, some courts in and outside of the United States are less willing or unwilling to protect trade secrets. If any of Ocuphire’s trade secrets were to be lawfully obtained or independently developed by a competitor, Ocuphire would have no right to prevent them, or those to whom they disclose such trade secrets, from using that technology or information to compete with it. If any of Ocuphire’s trade secrets were to be disclosed to, or independently developed by, a competitor or other third party, Ocuphire’s competitive position would be harmed.
Obtaining and maintaining Ocuphire’s trademark protection depends on approval from the USPTO and other foreign government agencies, and third parties may challenge, infringe, or otherwise weaken Ocuphire’s trademark rights.
Ocuphire has obtained registration of the “Nyxol” trademark in the United States. It has not yet registered trademarks for any other product candidates in any jurisdiction. If Ocuphire does not secure and maintain registrations for its trademarks, it may encounter more difficulty in enforcing them against third parties than it otherwise would, which could affect it business. When Ocuphire files trademark applications for a product candidate,
38
those applications may not be allowed for registration, and registered trademarks may not be obtained, maintained, or enforced. During trademark registration proceedings in the United States and foreign jurisdictions, Ocuphire may receive rejections. Ocuphire is given an opportunity to respond to those rejections, but may not be able to overcome such rejections. In addition, the USPTO and comparable agencies in many foreign jurisdictions allow third parties opportunities to oppose pending trademark applications and to seek to cancel registered trademarks. Opposition or cancellation proceedings may be filed against Ocuphire’s trademarks and its trademarks may not survive such proceedings.
In addition, any proprietary name Ocuphire proposes to use with a future product candidate in the United States must be approved by the FDA, regardless of whether Ocuphire has registered it, or applied to register it, as a trademark. The FDA typically conducts a review of proposed drug names, including an evaluation of potential for confusion with other drug names. If the FDA objects to any proposed proprietary drug name for any product candidate, Ocuphire may be required to expend significant additional resources in an effort to identify a suitable substitute proprietary drug name that would qualify under applicable trademark laws, not infringe the existing rights of third parties, and be acceptable to the FDA.
If Ocuphire registers any of its trademarks, its trademarks or trade names may be challenged, infringed, circumvented, declared generic, or determined to infringe on other marks. Ocuphire may not be able to protect its rights to these trademarks and trade names or may be forced to stop using these names, which Ocuphire needs for name recognition by potential partners or customers in its markets of interest. If Ocuphire is unable to establish name recognition based on its trademarks and trade names, Ocuphire may not be able to compete effectively and its business may be adversely affected.
Obtaining and maintaining Ocuphire’s patent protection depends on compliance with various procedural, document submission, fee payment, and other requirements imposed by governmental agencies, and its patent protection could be reduced or eliminated for noncompliance with these requirements.
The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment or other provisions during the patent application process. In addition, periodic maintenance and annuity fees on any issued patent are due to be paid to the USPTO and foreign patent agencies in several stages over the lifetime of the patent. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. In such an event, Ocuphire’s competitors might be able to enter the market, which would have an adverse effect on Ocuphire’s business.
Ocuphire depends on intellectual property sublicensed from Apexian Pharmaceuticals, Inc. (“Apexian”) for its APX3330 product candidate under development and its additional pipeline candidates, and the termination of, or reduction or loss of rights under, this sublicense would harm Ocuphire’s business.
Ocuphire entered into a sublicense agreement with Apexian (as amended, the “Apexian Sublicense Agreement”) to in-license intellectual property relating to the APX3330 product candidate and second generation product candidates, including certain study reports, manufacturing and analytical records, data, know-how, technical and other proprietary information relating to APX3330 that Apexian in-licensed from Eisai Co., Ltd. (“Eisai”). The rights granted under the Apexian Sublicense Agreement are subject to various milestone payment, royalty, insurance or other obligations on Ocuphire, and may be revocable under certain circumstances including if Ocuphire ceases to do business, fails to make the payments due thereunder, commits a material breach of the agreement that is not cured within a certain time period after receiving written notice or fails to meet certain specified development and commercial timelines. Additionally, if Ocuphire does not list its shares on a major stock exchange prior to December 31, 2020, either party may terminate the Apexian Sublicense Agreement. Termination of the Apexian Sublicense Agreement may result in Ocuphire having to negotiate a new or reinstated agreement, which may not be available to Ocuphire on equally favorable terms, or at all, which may mean Ocuphire is unable to develop or commercialize APX3330 and second generation assets.
Ocuphire does not have total control over the preparation, filing, prosecution and maintenance of patents and patent applications covering the technology that it licenses under the Apexian Sublicense Agreement. Under the Sublicense Agreement, Indiana University Research and Technology Corp. (“IURTC”), the owner of the patents licensed to Apexian and sublicensed to Ocuphire, maintains the right to control all prosecution and maintenance of
39
such patents. Therefore, Ocuphire cannot always be certain that these patents and patent applications will be prepared, filed, prosecuted and maintained in a manner consistent with the best interests of Ocuphire’s business. Although Ocuphire has a right to have its comments considered in connection with, and has agreed to bear the costs of, the prosecution and maintenance of the licensed patents, if IURTC fails to prosecute and maintain such patents, or loses rights to those patents or patent applications as a result of its control of the prosecution activities, the rights Ocuphire has licensed may be reduced or eliminated, and Ocuphire’s right to develop and commercialize any of its product candidates that are the subject of such licensed rights could be adversely affected.
Further, if Apexian breaches its license agreement with IURTC and fails to cure such breach within a 60-day cure period, IURTC may terminate such license agreement with Apexian, in which case, Ocuphire’s license shall also terminate and Ocuphire will lose all rights under the license agreement with Apexian. While the Apexian Sublicense Agreement provides that Apexian must cooperate with Ocuphire to remedy and cure Apexian’s breach of the license agreement with IURTC in order to prevent the termination of such license agreement, Ocuphire cannot guarantee that such efforts will be successful in preventing the termination of the license agreement between Apexian and IURTC. Similarly, if Apexian breaches its license agreement with Eisai and fails to cure such breach within a 60-day cure period, Eisai may terminate such license agreement with Apexian, in which case, Ocuphire’s sublicense rights under such license shall also terminate. While Ocuphire does not have any material obligations under the license agreement between Eisai and Apexian, Apexian has certain confidentiality and payment obligations that, if not met, could result in breach of the Eisai license agreement.
Under Apexian’s license agreement with IURTC, any act or omission by Ocuphire that would be a breach of the license agreement with IURTC if imputed to Apexian is deemed to be a breach by Apexian of such license agreement and cause for termination, including, in particular, any breach by Ocuphire of its payment, reporting, audit, and indemnification obligations.
The Apexian Sublicense Agreement obligates Ocuphire to make certain milestone payments.
Ocuphire is obligated to pay certain milestone payments to Apexian pursuant to the Apexian Sublicense Agreement. These milestone payments include (i) payments for specified developmental and regulatory milestones totaling up to $11 million in the aggregate and (ii) payments for specified sales milestones of up to $20 million in the aggregate.
Because certain of the milestone payments payable by Ocuphire are due upon certain events related to the development and regulatory approval of its product candidates, Ocuphire may be required to make such payments prior to the time at which it is able to generate revenue, if any, from sales any of its product candidates, if approved. There can be no assurance that Ocuphire will have the funds necessary to make such payments, or be able to raise such funds when needed, on terms acceptable to Ocuphire, or at all. Furthermore, if Ocuphire is forced to raise additional funds, it may be required to delay, limit, reduce or terminate its product development or future commercialization efforts, or grant rights to develop and market product candidates that it would otherwise develop and market themselves. If Ocuphire is unable to raise additional funds or maintain sufficient liquidity to make its payment obligations if and when they become due, it may be in material breach of its license and acquisition agreements and its counterparties may seek legal action or remedies against Ocuphire, which would harm its business, financial condition, results of operations and prospects.
Ocuphire may enter into collaborations, in-licensing arrangements, joint ventures, strategic alliances or partnerships with third-parties that may not result in the development of commercially viable products or the generation of significant future revenues.
Ocuphire may enter into certain license or other collaboration agreements in the future. Such agreements may impose various diligence, milestone payment, royalty, insurance or other obligations on Ocuphire. If Ocuphire fails to comply with such obligations, Ocuphire’s licensor or collaboration partners may have the right to terminate the relevant agreement, in which event Ocuphire would not be able to develop or market the products covered by such licensed intellectual property. Moreover, disputes may arise regarding intellectual property subject to a licensing agreement, including:
• |
the scope of rights granted under the license agreement and other interpretation-related issues; |
• |
the extent to which Ocuphire’s product candidates, technology and processes infringe on intellectual property of the licensor that is not subject to the licensing agreement; |
• |
the sublicensing of patent and other rights under Ocuphire’s collaborative development relationships; |
40
• |
Ocuphire’s diligence obligations under the license agreement and what activities satisfy those diligence obligations; |
• |
the inventorship and ownership of inventions and know-how resulting from the joint creation or use of intellectual property; and |
• |
the priority of invention of patented technology. |
In addition, the agreements under which intellectual property or technology is licensed from third parties are complex, and certain provisions in such agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation disagreement that may arise could narrow what Ocuphire believes to be the scope of Ocuphire’s rights to the relevant intellectual property or technology, or increase what Ocuphire believes to be Ocuphire’s financial or other obligations under the relevant agreement, either of which could have a material adverse effect on Ocuphire’s business, financial condition, results of operations, and prospects. Moreover, if disputes over intellectual property that Ocuphire has licensed prevent or impair Ocuphire’s ability to maintain Ocuphire’s licensing arrangements on commercially acceptable terms, Ocuphire may be unable to successfully develop and commercialize the affected product candidates, which could have a material adverse effect on Ocuphire’s business, financial conditions, results of operations, and prospects.
In addition, Ocuphire cannot be certain that the preparation, filing, prosecution and maintenance activities by any future licensors have been or will be conducted in compliance with applicable laws and regulations or will result in valid and enforceable patents and other intellectual property rights.
Risks Related to Ocuphire’s Employee Matters and Managing Growth
Ocuphire is dependent on its key personnel, and if it is not successful in attracting and retaining highly qualified personnel, it may not be able to successfully implement its business strategy.
Ocuphire is highly dependent on its management, scientific, and medical personnel, including Mina Sooch, its President, Chief Executive Officer and Board Vice Chair. Ocuphire has entered into employment agreements with its executive officers, but any employee may terminate his or her employment with Ocuphire. The loss of the services of any of Ocuphire’s executive officers, other key employees or consultants, or other scientific and medical advisors in the foreseeable future might impede the achievement of Ocuphire’s research, development, and commercialization objectives. Ocuphire relies on consultants and advisors, including scientific and clinical advisors, to assist it in formulating its development and commercialization strategy. Ocuphire’s consultants and advisors may be employed by employers other than Ocuphire and may have commitments under consulting or advisory contracts with other entities that may limit their availability to Ocuphire. Recruiting and retaining qualified scientific personnel and business and commercial personnel will also be critical to Ocuphire’s success. Ocuphire may not be able to attract and retain these personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for similar personnel. Ocuphire also experiences competition for the hiring of scientific personnel from universities and research institutions. Failure to succeed in clinical trials may also make it more challenging to recruit and retain qualified scientific personnel.
Ocuphire will need to develop and expand its company, and may encounter difficulties in managing this development and expansion, which could disrupt its operations.
As of February 1, 2021, Ocuphire had five full-time employees, and Ocuphire expects to increase its number of employees and the scope of its operations as it furthers the clinical development of its product candidates and becomes a public company. To manage its anticipated development and expansion, Ocuphire must continue to implement and improve its managerial, operational, and financial systems, expand its facilities, and continue to recruit and train additional qualified personnel. Also, Ocuphire’s management may need to divert a disproportionate amount of its attention away from its day-to-day activities and devote a substantial amount of time to managing these development activities. Due to its limited resources, Ocuphire may not be able to effectively manage the expansion of its operations or recruit and train additional qualified personnel. This may result in weaknesses in Ocuphire’s infrastructure, and give rise to operational mistakes, loss of business opportunities, loss of employees, or reduced productivity among remaining employees. The physical expansion of Ocuphire’s operations may lead to significant costs and may divert financial resources from other projects, such as the development of product candidates. If Ocuphire’s management is unable to effectively manage its expected development and expansion, its expenses may increase more than expected, its ability to generate or increase its revenue could be reduced and it may not be able
41
to implement its business strategy. Ocuphire’s future financial performance and its ability to commercialize product candidates, if approved, and compete effectively will depend, in part, on its ability to effectively manage the future development and expansion of Ocuphire.
A variety of risks associated with operating internationally for Ocuphire and its collaborators could adversely affect its business.
In addition to its U.S. operations, Ocuphire may pursue international operations in the future and would face risks associated with such global operations, including possible unfavorable regulatory, pricing and reimbursement, legal, political, tax, and labor conditions, which could harm its business. Ocuphire plans to conduct clinical trials outside of the United States. Ocuphire is subject to numerous risks associated with international business activities, including:
• |
compliance with differing or unexpected regulatory requirements for its product candidates; |
• |
different medical practices and customs affecting acceptance of its product candidates, if approved, or any other approved product in the marketplace; |
• |
language barriers; |
• |
the interpretation of contractual provisions governed by foreign law in the event of a contract dispute; |
• |
difficulties in staffing and managing foreign operations, and an inability to control commercial or other activities where it is relying on third parties; |
• |
workforce uncertainty in countries where labor unrest is more common than in the United States; |
• |
potential liability under the Foreign Corrupt Practice Act of 1977 or comparable foreign regulations; |
• |
production shortages resulting from any events affecting raw material supply or manufacturing capability abroad; |
• |
foreign government taxes, regulations, and permit requirements; |
• |
U.S. and foreign government tariffs, trade restrictions, price and exchange controls, and other regulatory requirements; |
• |
economic weakness, including inflation, natural disasters, war, events of terrorism, or political instability in particular foreign countries; |
• |
fluctuations in currency exchange rates, which could result in increased operating expenses and reduced revenues; |
• |
compliance with tax, employment, immigration, and labor laws, regulations, and restrictions for employees living or traveling abroad; |
• |
changes in diplomatic and trade relationships; and |
• |
challenges in enforcing its contractual and intellectual property rights, especially in those foreign countries that do not respect and protect intellectual property rights to the same extent as the United States. |
The COVID-19 pandemic has and could continue to adversely impact Ocuphire’s business, including pre-clinical and clinical trials and regulatory approvals.
On March 11, 2020, the World Health Organization declared the outbreak of COVID-19 as a global pandemic, which continues to spread throughout the United States and around the world. As a result of the COVID-19 pandemic, Ocuphire has experienced a few disruptions in its manufacturing, supply chain, research and development operations, regulatory process, and financial position. These disruptions have included the acceleration of shipment of active pharmaceutical ingredient supply from Italy and India, the convening of an FDA End-of-Phase 2 meeting via teleconference, and difficulties in obtaining more favorable financing terms. The global outbreak of COVID-19 continues to rapidly evolve. The extent to which the COVID-19 pandemic may impact Ocuphire’s business and pre-clinical and clinical trials will depend on future developments, which are highly uncertain and cannot be predicted with confidence, such as the duration of the outbreak, travel restrictions and social distancing in the U.S. and other countries, business closures or business disruptions and the effectiveness of actions taken in the U.S. and other countries to contain and treat the disease.
42
The COVID-19 pandemic poses the risk that Ocuphire, its employees, contractors, suppliers, and other partners may be prevented from conducting business activities for an indefinite period of time due to shutdowns that may be requested or mandated by state and federal governmental authorities. As COVID-19 continues to spread around the globe, Ocuphire may experience disruptions that could severely impact its business and planned clinical trials, including:
• |
interruption in global manufacturing and shipping that has affected, and may continue to affect the transport of clinical trial materials and materials, including testing equipment and personal protective equipment; |
• |
changes in local regulations as part of a response to the COVID-19 coronavirus outbreak which may result in unexpected costs; |
• |
delay in the timing of interactions with the FDA due to absenteeism by federal employees or by the diversion of their efforts and attention to approval of other therapeutics or other activities related to COVID-19; |
• |
impacts on Ocuphire’s ability to secure additional financing on favorable terms; and |
• |
modifications to the Ocuphire convertible notes. |
In addition, the outbreak of COVID-19 could disrupt Ocuphire’s operations due to absenteeism by infected or ill members of management or other employees, or absenteeism by members of management and other employees who elect not to come to work due to the illness affecting others in Ocuphire’s office or laboratory facilities, or due to quarantines. COVID-19 illness could also impact members of the Ocuphire Board and its ability to hold meetings. Although Ocuphire cannot estimate the length or gravity of the impact of the COVID-19 outbreak at this time, if the pandemic continues, it may have a material adverse effect on Ocuphire’s results of future operations, financial position, and liquidity over the next 12 or more months.
Ocuphire’s business and operations would suffer in the event of system failures or unplanned events.
Despite the implementation of security measures, Ocuphire’s internal computer systems and those of its current and future contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war, and telecommunications and electrical failures. While Ocuphire is not aware of any such material system failure, accident, or security breach to date, if such an event were to occur and cause interruptions in its operations, it could result in a material disruption of Ocuphire’s development programs and its business operations. For example, the loss of clinical trial data from completed or future clinical trials could result in delays in Ocuphire’s regulatory approval efforts and significantly increase Ocuphire’s costs to recover or reproduce the data. To the extent that any disruption or security breach were to result in a loss of, or damage to, Ocuphire’s data or applications, or inappropriate disclosure of confidential or proprietary information, Ocuphire could incur liability and the further development and commercialization of its product candidates could be delayed.
Furthermore, any unplanned event, such as flood, fire, explosion, tornadoes, earthquake, extreme weather condition, medical epidemics, power shortage, telecommunications failure, other natural or manmade accidents or incidents, or pandemics, including the ongoing COVID-19 pandemic, that result in Ocuphire being unable to fully utilize the facilities, may have an adverse effect on Ocuphire’s ability to operate its business, particularly on a daily basis, and have significant negative consequences on its financial and operating conditions. Loss of access to these facilities may result in increased costs, delays in the development of its product candidates, or interruption of its business operations.
Ocuphire’s insurance policies are expensive and protect only from some business risk, which leaves Ocuphire exposed to significant uninsured liabilities.
Ocuphire does not carry insurance for all categories of risks that its business may encounter, and insurance coverage is becoming increasingly expensive. Ocuphire does not know if it will be able to maintain existing insurance with adequate levels of coverage, and any liability insurance coverage it acquires in the future may not be sufficient to reimburse the company for any expenses or losses it may suffer. If Ocuphire obtains marketing approval for any product candidates that it may develop, Ocuphire intends to acquire insurance coverage to include the sale of commercial products, but it may be unable to obtain such insurance on commercially reasonable terms or in adequate amounts. Required coverage limits for such insurances are difficult to predict and may not be sufficient. If potential losses exceed Ocuphire’s insurance coverage, its financial condition would be adversely affected. In the event of contamination or injury, Ocuphire could be held liable for damages or be penalized with fines in an amount exceeding its resources. Clinical trials or regulatory approvals for any of its product candidates could be suspended, which could
43
adversely affect Ocuphire’s results of operations and business, including by preventing or limiting the development and commercialization of any product candidates that the company or its collaborators may develop.
In addition, as a public company, it may be more difficult or more costly for us to obtain certain types of insurance, including directors’ and officers’ liability insurance, and we may be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. The impact of these events could also make it more difficult for us to attract and retain qualified personnel to serve on our board of directors, our board committees or as executive officers.
Risks Related to Ownership of Ocuphire Common Stock
Ocuphire does not anticipate paying any cash dividends in the foreseeable future.
The current expectation is that Ocuphire will retain its future earnings, if any, to fund the development and growth of its business. As a result, capital appreciation, if any, of its common stock will be your sole source of gain, if any, for the foreseeable future.
If Ocuphire fails to comply with the continued listing standards of the Nasdaq Capital Market, Ocuphire common stock could be delisted. If it is delisted, Ocuphire common stock and the liquidity of its common stock would be impacted.
The continued listing of Ocuphire common stock on Nasdaq is contingent on Ocuphire's continued compliance with a number of listing standards. There is no assurance that Ocuphire will remain in compliance with these standards. Delisting from Nasdaq would adversely affect Ocuphire's ability to raise additional financing through the public or private sale of equity securities, significantly affect the ability of investors to trade Ocuphire's securities and negatively affect the value and liquidity of Ocuphire's common stock. Delisting also could limit Ocuphire's strategic alternatives and attractiveness to potential counterparties and have other negative results, including the potential loss of employee confidence, the loss of institutional investors or interest in business development opportunities. Moreover, Ocuphire committed in connection with the sale of securities to use commercially reasonable efforts to maintain the listing of its common stock during such time that certain warrants are outstanding.
The market price of Ocuphire common stock may fluctuate significantly.
The market price of Ocuphire common stock may fluctuate significantly in response to factors, some of which are beyond Ocuphire's control, such as:
• |
the announcement of new products or product enhancements by Ocuphire or its competitors; |
• |
changes in Ocuphire's relationships with its licensors or other strategic partners; |
• |
developments concerning intellectual property rights and regulatory approvals; |
• |
variations in Ocuphire's and Ocuphire's competitors’ results of operations; |
• |
substantial sales of shares of our common stock due to the release of lock-up agreements; |
• |
the announcement of clinical trial results; |
• |
the announcement of potentially dilutive financings; |
• |
changes in earnings estimates or recommendations by securities analysts; |
• |
changes in the structure of healthcare payment systems; and |
• |
developments and market conditions in the pharmaceutical and biotechnology industries, including due to the COVID-19 pandemic. |
44
Further, the stock market, in general, and the market for biotechnology companies, in particular, have experienced extreme price and volume fluctuations. Continued market fluctuations could result in extreme volatility in the price of Ocuphire's common stock, which may be unrelated or disproportionate to Ocuphire's operating performance and which could cause a decline in the value of Ocuphire's common stock. You should also be aware that price volatility might be worse if the trading volume of Ocuphire common stock is low.
Ocuphire may be subject to securities litigation, which is expensive and could divert management attention.
The market price of Ocuphire common stock may be volatile, and in the past companies that have experienced volatility in the market price of their stock have been subject to securities class action litigation. Ocuphire may be the target of this type of litigation in the future. Securities litigation against Ocuphire could result in substantial costs and direct Ocuphire management’s attention from other business concerns, which could seriously harm Ocuphire's business.
45
This prospectus and the documents we have filed with the SEC that are incorporated by reference contain “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended, or the Exchange Act. These statements relate to future events or to our future operating or financial performance and involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performances or achievements expressed or implied by the forward-looking statements. All statements other than statements of historical facts contained in this prospectus, including statements regarding our future results of operations and financial condition, business strategy and plans and objectives of management for future operations, are forward-looking statements. In some cases, forward-looking statements may be identified by words such as “anticipate,” “believe,” “continue,” “could,” “design,” “estimate,” “expect,” “intend,” “may,” “plan,” “potentially,” “predict,” “project,” “should,” “will” or the negative of these terms or other similar expressions.
We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. These forward-looking statements are subject to a number of known and unknown risks, uncertainties and assumptions, including risks described in the section titled “Risk Factors” and elsewhere in this prospectus and in our most recent Annual Report on Form 10-K and Quarterly Report on Form 10-Q, as well as any amendments thereto reflected in subsequent filings with the SEC, which are incorporated by reference into this prospectus in their entirety, together with other information in this prospectus, the documents incorporated by reference and any free writing prospectus that we may authorize for use in connection with a specific offering. These factors include, among other things:
• |
We may never earn a profit; |
• |
We are subject to the uncertainties associated with the clinical development and regulatory approval of its product candidates including potential delays in the commencement, enrollment and completion of clinical trials and that the results of prior clinical trials may not be predictive of future results; |
• |
We will be required to raise additional funds to finance its operations and remain a going concern and may be required to do so sooner than it expects; |
• |
We may not be able to raise additional funds when necessary, and/or on acceptable terms; |
• |
We may not be able to protect our respective intellectual property rights; |
• |
There may be changes in expected or existing competition for our product candidates; |
• |
if we were to be delisted from Nasdaq, it could reduce the visibility, liquidity and price of its common stock; |
• |
a significant portion of the our total outstanding shares of common stock may be sold into the public market at any point, which could cause the market price of our common stock to drop significantly, even if we are doing well; |
• |
we do not anticipate paying any cash dividends on its capital stock in the foreseeable future; |
• |
provisions in our certificate of incorporation, our bylaws or Delaware law might discourage, delay or prevent a change in control of the company or changes in its management, which may depress the price of its common stock; |
• |
the coronavirus (COVID-19) pandemic may have an adverse effect on our business, the medical community and the global economy; and |
• |
securities analysts’ published reports could cause a decline in the price of the our stock. |
These risks are not exhaustive. Other sections of this prospectus may include additional factors that could harm our business and financial performance. Moreover, we operate in a very competitive and rapidly changing environment.
46
New risks and uncertainties emerge from time to time, and it is not possible for our management to predict all risks and uncertainties nor can we assess the impact of all such factors on our business or the extent to which any such factor, or combination of such factors, may cause actual results to differ from those contained in, or implied by, any forward-looking statements.
You should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. Except as required by law, we undertake no obligation to update publicly any forward-looking statements for any reason after the date of this prospectus or to conform these statements to actual results or to changes in our expectations.
In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this prospectus, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements.
You should read this prospectus and the documents that we reference in this prospectus and have filed as exhibits to the registration statement of which this prospectus is a part with the understanding that our actual future results, levels of activity, performance and achievements may be different from what we expect. We qualify all of our forward-looking statements by these cautionary statements.
47
Except as described in any applicable prospectus supplement or in any free writing prospectuses we have authorized for use in connection with a specific offering, we currently intend to use the net proceeds from this offering for working capital and general corporate purposes, which may include, among other things, funding research and development programs, vendor payables, hiring additional personnel, and capital expenditures.
We will not receive any proceeds from the sale of shares of our common stock by any selling stockholders.
48
General
As of the date of this prospectus, our certificate of incorporation authorizes us to issue up to 85,000,000 shares of capital stock, all with a par value of $0.0001 per share, of which: 75,000,000 shares are designated as common stock and 10,000,000 shares are designated as preferred stock.
The following summary describes the material terms of our capital stock. The description of capital stock is qualified by reference to our certificate of incorporation and our bylaws.
Common Stock
As of December 31, 2020, 10,882,495 shares of common stock were outstanding. All outstanding shares of common stock are duly authorized, validly issued, fully paid, and nonassessable. All authorized but unissued shares of our common stock are available for issuance by our board of directors without any further stockholder action, except as required by the listing standards of Nasdaq.
Voting Rights
Holders of our common stock are entitled to one vote for each share held on all matters submitted to a vote of stockholders, including the election of directors, and do not have cumulative voting rights. Accordingly, the holders of a majority of the outstanding shares of common stock entitled to vote in any election of directors can elect all of the directors standing for election, if they so choose, other than any directors that holders of any preferred stock we may issue may be entitled to elect.
Dividend Rights
Subject to preferences that may be applicable to any then-outstanding preferred stock, holders of common stock are entitled to receive ratably those dividends, if any, as may be declared by the board of directors out of legally available funds.
Liquidation
In the event of our liquidation, dissolution, or winding up, the holders of common stock will be entitled to share ratably in the assets legally available for distribution to stockholders after the payment of or provision for all of our debts and other liabilities, subject to the prior rights of any preferred stock then-outstanding.
Rights and Preferences
Holders of common stock have no preemptive or conversion rights or other subscription rights and there are no redemption or sinking fund provisions applicable to the common stock. The rights, preferences, and privileges of holders of common stock are subject to and may be adversely affected by the rights of the holders of shares of any series of preferred stock that we may designate and issue in the future.
Preferred Stock
As of December 31, 2020, no shares of preferred stock were outstanding. Our board of directors may, without further action by our stockholders, fix the rights, preferences, privileges and restrictions of up to an aggregate of 10,000,000 shares of preferred stock in one or more series and authorize their issuance. These rights, preferences and privileges could include dividend rights, conversion rights, voting rights, terms of redemption, liquidation preferences, sinking fund terms and the number of shares constituting any series or the designation of such series, any or all of which may be greater than the rights of our common stock. The issuance of our preferred stock could adversely affect the voting power of holders of our common stock and the likelihood that such holders will receive dividend payments and payments upon liquidation. In addition, the issuance of preferred stock could have the effect of decreasing the market price of our common stock and could also have the effect of delaying, deferring or preventing a change of control or other corporate action.
49
We will fix the designations, voting powers, preferences and rights of the preferred stock of each series we issue under this prospectus, as well as the qualifications, limitations or restrictions thereof, in the certificate of designation relating to that series. We will file as an exhibit to the registration statement of which this prospectus is a part, or will incorporate by reference from reports that we file with the SEC, the form of any certificate of designation that describes the terms of the series of preferred stock we are offering. We will describe in the applicable prospectus supplement the terms of the series of preferred stock being offered, including, to the extent applicable:
• |
the title and stated value; |
• |
the number of shares we are offering; |
• |
the liquidation preference per share; |
• |
the purchase price; |
• |
the dividend rate, period and payment date and method of calculation for dividends; |
• |
whether dividends will be cumulative or non-cumulative and, if cumulative, the date from which dividends will accumulate; |
• |
the procedures for any auction and remarketing; |
• |
the provisions for a sinking fund; |
• |
the provisions for redemption or repurchase, if applicable, and any restrictions on our ability to exercise those redemption and repurchase rights; |
• |
any listing of the preferred stock on any securities exchange or market; |
• |
whether the preferred stock will be convertible into our common stock, and, if applicable, the conversion price, or how it will be calculated, and the conversion period; |
• |
whether the preferred stock will be exchangeable into debt securities, and, if applicable, the exchange price, or how it will be calculated, and the exchange period; |
• |
voting rights of the preferred stock; |
• |
preemptive rights; |
• |
restrictions on transfer, sale or other assignment; |
• |
whether interests in the preferred stock will be represented by depositary shares; |
• |
a discussion of material United States federal income tax considerations applicable to the preferred stock; |
• |
the relative ranking and preferences of the preferred stock as to dividend rights and rights if we liquidate, dissolve or wind up our affairs; |
• |
any limitations on the issuance of any class or series of preferred stock ranking senior to or on a parity with the series of preferred stock as to dividend rights and rights if we liquidate, dissolve or wind up our affairs; and |
• |
any other specific terms, preferences, rights or limitations of, or restrictions on, the preferred stock. |
If we issue shares of preferred stock under this prospectus, the shares will be fully paid and non-assessable.
Options and Restricted Stock
As of December 31, 2020, (i) stock options to purchase an aggregate of 583,500 shares of common stock were outstanding under our 2020 Equity Incentive Plan, or 2020 Plan, stock options to purchase an aggregate of 1,200,698 shares of common stock were outstanding under our 2018 Equity Incentive Plan, or 2018 Plan, and 123 shares of common stock were outstanding under our 2003 Stock Option Plan, or 2003 Plan, and (ii) 40,000 unvested shares of restricted stock were outstanding.
Warrants
As of December 31, 2020, 6,398,212 warrants to purchase shares of our capital stock were outstanding, with a weighted average exercise price of $4.52 (subject to adjustment).
50
Anti-Takeover Effects of Delaware Law and Our Certificate of Incorporation and Bylaws
Some provisions of Delaware law, our amended and restated certificate of incorporation, and our amended and restated bylaws contain provisions that could make the following transactions more difficult: an acquisition of us by means of a tender offer; an acquisition of us by means of a proxy contest or otherwise; or the removal of our incumbent officers and directors. It is possible that these provisions could make it more difficult to accomplish or could deter transactions that stockholders may otherwise consider to be in their best interest or in our best interests, including transactions which provide for payment of a premium over the market price for our shares.
These provisions, summarized below, are intended to discourage coercive takeover practices and inadequate takeover bids. These provisions are also designed to encourage persons seeking to acquire control of us to first negotiate with our board of directors. We believe that the benefits of the increased protection of our potential ability to negotiate with the proponent of an unfriendly or unsolicited proposal to acquire or restructure us outweigh the disadvantages of discouraging these proposals because negotiation of these proposals could result in an improvement of their terms.
Stockholder Meetings
Our bylaws provide that a special meeting of stockholders may be called only by our chairperson of the board, chief executive officer or president, or by a resolution adopted by a majority of our board of directors.
Requirements for Advance Notification of Stockholder Nominations and Proposals
Our bylaws establish advance notice procedures with respect to stockholder proposals to be brought before a stockholder meeting and the nomination of candidates for election as directors, other than nominations made by or at the direction of the board of directors or a committee of the board of directors.
Elimination of Stockholder Action by Written Consent
Our bylaws eliminate the right of stockholders to act by written consent without a meeting.
Removal of Directors
Our certificate of incorporation provides for the removal of any of its directors only for cause and requires a stockholder vote of at least a majority of the voting power of the then outstanding voting stock.
Stockholders Not Entitled to Cumulative Voting
Our certificate of incorporation does not permit stockholders to cumulate their votes in the election of directors. Accordingly, the holders of a majority of the outstanding shares of our common stock entitled to vote in any election of directors can elect all of the directors standing for election, if they choose, other than any directors that holders of our preferred stock may be entitled to elect.
Delaware Anti-Takeover Statute
We are subject to Section 203 of the Delaware General Corporation Law, or DGCL, which prohibits persons deemed to be “interested stockholders” from engaging in a “business combination” with a publicly held Delaware corporation for three years following the date these persons become interested stockholders unless the business combination is, or the transaction in which the person became an interested stockholder was, approved in a prescribed manner or another prescribed exception applies. Generally, an “interested stockholder” is a person who, together with affiliates and associates, owns, or within three years prior to the determination of interested stockholder status did own, 15% or more of a corporation’s voting stock. Generally, a “business combination” includes a merger, asset or stock sale, or other transaction resulting in a financial benefit to the interested stockholder. The existence of this provision may have an anti-takeover effect with respect to transactions not approved in advance by the board of directors.
The provisions of Delaware law, our certificate of incorporation, and our bylaws could have the effect of discouraging others from attempting hostile takeovers and, as a consequence, they may also inhibit temporary fluctuations in the market price of our common stock that often result from actual or rumored hostile takeover attempts. These provisions may also have the effect of preventing changes in the composition of our board and management. It is possible that these provisions could make it more difficult to accomplish transactions that stockholders may otherwise deem to be in their best interests.
51
Transfer Agent and Registrar
The transfer agent and registrar for our common stock is Olde Monmouth Stock Transfer Co.
Listing on the Nasdaq Capital Market
Our common stock is listed on the Nasdaq Capital Market under the symbol “OCUP.”
52
We may issue debt securities from time to time, in one or more series, as either senior or subordinated debt or as senior or subordinated convertible debt. While the terms we have summarized below will apply generally to any debt securities that we may offer under this prospectus, we will describe the particular terms of any debt securities that we may offer in more detail in the applicable prospectus supplement. The terms of any debt securities offered under a prospectus supplement may differ from the terms described below. Unless the context requires otherwise, whenever we refer to the indenture, we also are referring to any supplemental indentures that specify the terms of a particular series of debt securities.
We will issue the debt securities under the indenture that we will enter into with the trustee named in the indenture. The indenture will be qualified under the Trust Indenture Act of 1939, as amended, or the Trust Indenture Act. We have filed the form of indenture as an exhibit to the registration statement of which this prospectus is a part, and supplemental indentures and forms of debt securities containing the terms of the debt securities being offered will be filed as exhibits to the registration statement of which this prospectus is a part or will be incorporated by reference from reports that we file with the SEC.
The following summary of material provisions of the debt securities and the indenture is subject to, and qualified in its entirety by reference to, all of the provisions of the indenture applicable to a particular series of debt securities. We urge you to read the applicable prospectus supplements and any related free writing prospectuses related to the debt securities that we may offer under this prospectus, as well as the complete indenture that contains the terms of the debt securities.
General
The indenture does not limit the amount of debt securities that we may issue. It provides that we may issue debt securities up to the principal amount that we may authorize and may be in any currency or currency unit that we may designate. Except for the limitations on consolidation, merger and sale of all or substantially all of our assets contained in the indenture, the terms of the indenture do not contain any covenants or other provisions designed to give holders of any debt securities protection against changes in our operations, financial condition or transactions involving us.
We may issue the debt securities issued under the indenture as “discount securities,” which means they may be sold at a discount below their stated principal amount. These debt securities, as well as other debt securities that are not issued at a discount, may be issued with “original issue discount,” or OID, for U.S. federal income tax purposes because of interest payment and other characteristics or terms of the debt securities. Material U.S. federal income tax considerations applicable to debt securities issued with OID will be described in more detail in any applicable prospectus supplement.
We will describe in the applicable prospectus supplement the terms of the series of debt securities being offered, including:
• |
the title of the series of debt securities; |
• |
any limit upon the aggregate principal amount that may be issued; |
• |
the maturity date or dates; |
• |
the form of the debt securities of the series; |
• |
the applicability of any guarantees; |
• |
whether or not the debt securities will be secured or unsecured, and the terms of any secured debt; |
• |
whether the debt securities rank as senior debt, senior subordinated debt, subordinated debt or any combination thereof, and the terms of any subordination; |
• |
if the price (expressed as a percentage of the aggregate principal amount thereof) at which such debt securities will be issued is a price other than the principal amount thereof, the portion of the principal amount thereof payable upon declaration of acceleration of the maturity thereof, or if applicable, the portion of the principal amount of such debt securities that is convertible into another security or the method by which any such portion shall be determined; |
53
• |
the interest rate or rates, which may be fixed or variable, or the method for determining the rate and the date interest will begin to accrue, the dates interest will be payable and the regular record dates for interest payment dates or the method for determining such dates; |
• |
our right, if any, to defer payment of interest and the maximum length of any such deferral period; |
• |
if applicable, the date or dates after which, or the period or periods during which, and the price or prices at which, we may, at our option, redeem the series of debt securities pursuant to any optional or provisional redemption provisions and the terms of those redemption provisions; |
• |
the date or dates, if any, on which, and the price or prices at which we are obligated, pursuant to any mandatory sinking fund or analogous fund provisions or otherwise, to redeem, or at the holder’s option to purchase, the series of debt securities and the currency or currency unit in which the debt securities are payable; |
• |
the denominations in which we will issue the series of debt securities, if other than denominations of $1,000 and any integral multiple thereof; |
• |
any and all terms, if applicable, relating to any auction or remarketing of the debt securities of that series and any security for our obligations with respect to such debt securities and any other terms which may be advisable in connection with the marketing of debt securities of that series; |
• |
whether the debt securities of the series shall be issued in whole or in part in the form of a global security or securities; the terms and conditions, if any, upon which such global security or securities may be exchanged in whole or in part for other individual securities; and the depositary for such global security or securities; |
• |
if applicable, the provisions relating to conversion or exchange of any debt securities of the series and the terms and conditions upon which such debt securities will be so convertible or exchangeable, including the conversion or exchange price, as applicable, or how it will be calculated and may be adjusted, any mandatory or optional (at our option or the holders’ option) conversion or exchange features, the applicable conversion or exchange period and the manner of settlement for any conversion or exchange; |
• |
if other than the full principal amount thereof, the portion of the principal amount of debt securities of the series which shall be payable upon declaration of acceleration of the maturity thereof; |
• |
additions to or changes in the covenants applicable to the particular debt securities being issued, including, among others, the consolidation, merger or sale covenant; |
• |
additions to or changes in the events of default with respect to the securities and any change in the right of the trustee or the holders to declare the principal, premium, if any, and interest, if any, with respect to such securities to be due and payable; |
• |
additions to or changes in or deletions of the provisions relating to covenant defeasance and legal defeasance; |
• |
additions to or changes in the provisions relating to satisfaction and discharge of the indenture; |
• |
additions to or changes in the provisions relating to the modification of the indenture both with and without the consent of holders of debt securities issued under the indenture; |
• |
the currency of payment of debt securities if other than U.S. dollars and the manner of determining the equivalent amount in U.S. dollars; |
• |
whether interest will be payable in cash or additional debt securities at our or the holders’ option and the terms and conditions upon which the election may be made; |
• |
the terms and conditions, if any, upon which we will pay amounts in addition to the stated interest, premium, if any and principal amounts of the debt securities of the series to any holder that is not a “United States person” for federal tax purposes; |
• |
any restrictions on transfer, sale or assignment of the debt securities of the series; and |
• |
any other specific terms, preferences, rights or limitations of, or restrictions on, the debt securities, any other additions or changes in the provisions of the indenture, and any terms that may be required by us or advisable under applicable laws or regulations. |
54
Conversion or Exchange Rights
We will set forth in the applicable prospectus supplement the terms on which a series of debt securities may be convertible into or exchangeable for our common stock or our other securities. We will include provisions as to settlement upon conversion or exchange and whether conversion or exchange is mandatory, at the option of the holder or at our option. We may include provisions pursuant to which the number of shares of our common stock or our other securities that the holders of the series of debt securities receive would be subject to adjustment.
Consolidation, Merger or Sale
Unless we provide otherwise in the prospectus supplement applicable to a particular series of debt securities, the indenture will not contain any covenant that restricts our ability to merge or consolidate, or sell, convey, transfer or otherwise dispose of our assets as an entirety or substantially as an entirety. However, any successor to or acquirer of such assets (other than a subsidiary of ours) must assume all of our obligations under the indenture or the debt securities, as appropriate.
Events of Default under the Indenture
Unless we provide otherwise in the prospectus supplement applicable to a particular series of debt securities, the following are events of default under the indenture with respect to any series of debt securities that we may issue:
• |
if we fail to pay any installment of interest on any series of debt securities, as and when the same shall become due and payable, and such default continues for a period of 90 days; provided, however, that a valid extension of an interest payment period by us in accordance with the terms of any indenture supplemental thereto shall not constitute a default in the payment of interest for this purpose; |
• |
if we fail to pay the principal of, or premium, if any, on any series of debt securities as and when the same shall become due and payable whether at maturity, upon redemption, by declaration or otherwise, or in any payment required by any sinking or analogous fund established with respect to such series; provided, however, that a valid extension of the maturity of such debt securities in accordance with the terms of any indenture supplemental thereto shall not constitute a default in the payment of principal or premium, if any; |
• |
if we fail to observe or perform any other covenant or agreement contained in the debt securities or the indenture, other than a covenant specifically relating to another series of debt securities, and our failure continues for 90 days after we receive written notice of such failure, requiring the same to be remedied and stating that such is a notice of default thereunder, from the trustee or holders of at least 25% in aggregate principal amount of the outstanding debt securities of the applicable series; and |
• |
if specified events of bankruptcy, insolvency or reorganization occur. |
If an event of default with respect to debt securities of any series occurs and is continuing, other than an event of default specified in the last bullet point above, the trustee or the holders of at least 25% in aggregate principal amount of the outstanding debt securities of that series, by notice to us in writing, and to the trustee if notice is given by such holders, may declare the unpaid principal of, premium, if any, and accrued interest, if any, due and payable immediately. If an event of default specified in the last bullet point above occurs with respect to us, the principal amount of and accrued interest, if any, of each issue of debt securities then outstanding shall be due and payable without any notice or other action on the part of the trustee or any holder.
The holders of a majority in principal amount of the outstanding debt securities of an affected series may waive any default or event of default with respect to the series and its consequences, except defaults or events of default regarding payment of principal, premium, if any, or interest, unless we have cured the default or event of default in accordance with the indenture. Any waiver shall cure the default or event of default.
Subject to the terms of the indenture, if an event of default under an indenture shall occur and be continuing, the trustee will be under no obligation to exercise any of its rights or powers under such indenture at the request or direction of any of the holders of the applicable series of debt securities, unless such holders have offered the trustee reasonable indemnity. The holders of a majority in principal amount of the outstanding debt securities of any series will have the right to direct the time, method and place of conducting any proceeding for any remedy available to the trustee, or exercising any trust or power conferred on the trustee, with respect to the debt securities of that series, provided that:
• |
the direction so given by the holder is not in conflict with any law or the applicable indenture; and |
• |
subject to its duties under the Trust Indenture Act, the trustee need not take any action that might involve it in personal liability or might be unduly prejudicial to the holders not involved in the proceeding. |
55
A holder of the debt securities of any series will have the right to institute a proceeding under the indenture or to appoint a receiver or trustee, or to seek other remedies only if:
• |
the holder has given written notice to the trustee of a continuing event of default with respect to that series; |
• |
the holders of at least 25% in aggregate principal amount of the outstanding debt securities of that series have made written request; |
• |
such holders have offered to the trustee indemnity satisfactory to it against the costs, expenses and liabilities to be incurred by the trustee in compliance with the request; and |
• |
the trustee does not institute the proceeding, and does not receive from the holders of a majority in aggregate principal amount of the outstanding debt securities of that series other conflicting directions within 90 days after the notice, request and offer. |
These limitations do not apply to a suit instituted by a holder of debt securities if we default in the payment of the principal, premium, if any, or interest on, the debt securities.
We will periodically file statements with the trustee regarding our compliance with specified covenants in the indenture.
Modification of Indenture; Waiver
We and the trustee may change an indenture without the consent of any holders with respect to specific matters:
• |
to cure any ambiguity, defect or inconsistency in the indenture or in the debt securities of any series; |
• |
to comply with the provisions described above under “Description of Debt Securities—Consolidation, Merger or Sale;” |
• |
to provide for uncertificated debt securities in addition to or in place of certificated debt securities; |
• |
to add to our covenants, restrictions, conditions or provisions such new covenants, restrictions, conditions or provisions for the benefit of the holders of all or any series of debt securities, to make the occurrence, or the occurrence and the continuance, of a default in any such additional covenants, restrictions, conditions or provisions an event of default or to surrender any right or power conferred upon us in the indenture; |
• |
to add to, delete from or revise the conditions, limitations, and restrictions on the authorized amount, terms, or purposes of issue, authentication and delivery of debt securities, as set forth in the indenture; |
• |
to make any change that does not adversely affect the interests of any holder of debt securities of any series in any material respect; |
• |
to provide for the issuance of and establish the form and terms and conditions of the debt securities of any series as provided above under “Description of Debt Securities—General” to establish the form of any certifications required to be furnished pursuant to the terms of the indenture or any series of debt securities, or to add to the rights of the holders of any series of debt securities; |
• |
to evidence and provide for the acceptance of appointment under any indenture by a successor trustee; or |
• |
to comply with any requirements of the SEC in connection with the qualification of any indenture under the Trust Indenture Act. |
In addition, under the indenture, the rights of holders of a series of debt securities may be changed by us and the trustee with the written consent of the holders of at least a majority in aggregate principal amount of the outstanding debt securities of each series that is affected. However, unless we provide otherwise in the prospectus supplement applicable to a particular series of debt securities, we and the trustee may make the following changes only with the consent of each holder of any outstanding debt securities affected:
• |
extending the fixed maturity of any debt securities of any series; |
• |
reducing the principal amount, reducing the rate of or extending the time of payment of interest, or reducing any premium payable upon the redemption of any series of any debt securities; or |
• |
reducing the percentage of debt securities, the holders of which are required to consent to any amendment, supplement, modification or waiver. |
56
Discharge
Each indenture provides that we can elect to be discharged from our obligations with respect to one or more series of debt securities, except for specified obligations, including obligations to:
• |
provide for payment; |
• |
register the transfer or exchange of debt securities of the series; |
• |
replace stolen, lost or mutilated debt securities of the series; |
• |
pay principal of and premium and interest on any debt securities of the series; |
• |
maintain paying agencies; |
• |
hold monies for payment in trust; |
• |
recover excess money held by the trustee; |
• |
compensate and indemnify the trustee; and |
• |
appoint any successor trustee. |
In order to exercise our rights to be discharged, we must deposit with the trustee money or government obligations sufficient to pay all the principal of, any premium, if any, and interest on, the debt securities of the series on the dates payments are due.
Form, Exchange and Transfer
We will issue the debt securities of each series only in fully registered form without coupons and, unless we provide otherwise in the applicable prospectus supplement, in denominations of $1,000 and any integral multiple thereof. The indenture provides that we may issue debt securities of a series in temporary or permanent global form and as book-entry securities that will be deposited with, or on behalf of, The Depository Trust Company, or DTC, or another depositary named by us and identified in the applicable prospectus supplement with respect to that series. To the extent the debt securities of a series are issued in global form and as book-entry, a description of terms relating to any book-entry securities will be set forth in the applicable prospectus supplement.
At the option of the holder, subject to the terms of the indenture and the limitations applicable to global securities described in the applicable prospectus supplement, the holder of the debt securities of any series can exchange the debt securities for other debt securities of the same series, in any authorized denomination and of like tenor and aggregate principal amount.
Subject to the terms of the indenture and the limitations applicable to global securities set forth in the applicable prospectus supplement, holders of the debt securities may present the debt securities for exchange or for registration of transfer, duly endorsed or with the form of transfer endorsed thereon duly executed if so required by us or the security registrar, at the office of the security registrar or at the office of any transfer agent designated by us for this purpose. Unless otherwise provided in the debt securities that the holder presents for transfer or exchange, we will impose no service charge for any registration of transfer or exchange, but we may require payment of any taxes or other governmental charges.
We will name in the applicable prospectus supplement the security registrar, and any transfer agent in addition to the security registrar, that we initially designate for any debt securities. We may at any time designate additional transfer agents or rescind the designation of any transfer agent or approve a change in the office through which any transfer agent acts, except that we will be required to maintain a transfer agent in each place of payment for the debt securities of each series.
If we elect to redeem the debt securities of any series, we will not be required to:
• |
issue, register the transfer of, or exchange any debt securities of that series during a period beginning at the opening of business 15 days before the day of mailing of a notice of redemption of any debt securities that may be selected for redemption and ending at the close of business on the day of the mailing; or |
• |
register the transfer of or exchange any debt securities so selected for redemption, in whole or in part, except the unredeemed portion of any debt securities we are redeeming in part. |
57
Information Concerning the Trustee
The trustee, other than during the occurrence and continuance of an event of default under an indenture, undertakes to perform only those duties as are specifically set forth in the applicable indenture. Upon an event of default under an indenture, the trustee must use the same degree of care as a prudent person would exercise or use in the conduct of his or her own affairs. Subject to this provision, the trustee is under no obligation to exercise any of the powers given it by the indenture at the request of any holder of debt securities unless it is offered reasonable security and indemnity against the costs, expenses and liabilities that it might incur.
Payment and Paying Agents
Unless we otherwise indicate in the applicable prospectus supplement, we will make payment of the interest on any debt securities on any interest payment date to the person in whose name the debt securities, or one or more predecessor securities, are registered at the close of business on the regular record date for the interest.
We will pay principal of and any premium and interest on the debt securities of a particular series at the office of the paying agents designated by us, except that unless we otherwise indicate in the applicable prospectus supplement, we will make interest payments by check that we will mail to the holder or by wire transfer to certain holders. Unless we otherwise indicate in the applicable prospectus supplement, we will designate the corporate trust office of the trustee as our sole paying agent for payments with respect to debt securities of each series. We will name in the applicable prospectus supplement any other paying agents that we initially designate for the debt securities of a particular series. We will maintain a paying agent in each place of payment for the debt securities of a particular series.
All money we pay to a paying agent or the trustee for the payment of the principal of or any premium or interest on any debt securities that remains unclaimed at the end of two years after such principal, premium or interest has become due and payable will be repaid to us, and the holder of the debt security thereafter may look only to us for payment thereof.
Governing Law
The indenture and the debt securities will be governed by and construed in accordance with the internal laws of the State of New York, except to the extent that the Trust Indenture Act of 1939 is applicable.
58
The following description, together with the additional information we may include in any applicable prospectus supplement and free writing prospectus, summarizes the material terms and provisions of the warrants that we may offer under this prospectus, which may consist of warrants to purchase common stock, preferred stock or debt securities and may be issued in one or more series. Warrants may be offered independently or in combination with common stock, preferred stock or debt securities offered by any prospectus supplement. While the terms we have summarized below will apply generally to any warrants that we may offer under this prospectus, we will describe the particular terms of any series of warrants in more detail in the applicable prospectus supplement. The following description of warrants will apply to the warrants offered by this prospectus unless we provide otherwise in the applicable prospectus supplement. The applicable prospectus supplement for a particular series of warrants may specify different or additional terms.
We have filed forms of the warrant agreements and forms of warrant certificates containing the terms of the warrants that may be offered as exhibits to the registration statement of which this prospectus is a part. We will file as exhibits to the registration statement of which this prospectus is a part, or will incorporate by reference from reports that we file with the SEC, the form of warrant and/or the warrant agreement and warrant certificate, as applicable, that contain the terms of the particular series of warrants we are offering, and any supplemental agreements, before the issuance of such warrants. The following summaries of material terms and provisions of the warrants are subject to, and qualified in their entirety by reference to, all the provisions of the form of warrant and/or the warrant agreement and warrant certificate, as applicable, and any supplemental agreements applicable to a particular series of warrants that we may offer under this prospectus. We urge you to read the applicable prospectus supplement related to the particular series of warrants that we may offer under this prospectus, as well as any related free writing prospectus, and the complete form of warrant and/or the warrant agreement and warrant certificate, as applicable, and any supplemental agreements, that contain the terms of the warrants.
General
We will describe in the applicable prospectus supplement the terms of the series of warrants being offered, including:
• |
the offering price and aggregate number of warrants offered; |
• |
the currency for which the warrants may be purchased; |
• |
if applicable, the designation and terms of the securities with which the warrants are issued and the number of warrants issued with each such security or each principal amount of such security; |
• |
in the case of warrants to purchase debt securities, the principal amount of debt securities purchasable upon exercise of one warrant and the price at, and currency in which, this principal amount of debt securities may be purchased upon such exercise; |
• |
in the case of warrants to purchase common stock or preferred stock, the number of shares of common stock or preferred stock, as the case may be, purchasable upon the exercise of one warrant and the price at which these shares may be purchased upon such exercise; |
• |
the effect of any merger, consolidation, sale or other disposition of our business on the warrant agreements and the warrants; |
• |
the terms of any rights to redeem or call the warrants; |
• |
any provisions for changes to or adjustments in the exercise price or number of securities issuable upon exercise of the warrants; |
• |
the dates on which the right to exercise the warrants will commence and expire; |
• |
the manner in which the warrant agreements and warrants may be modified; |
• |
a discussion of any material or special U.S. federal income tax considerations of holding or exercising the warrants; |
• |
the terms of the securities issuable upon exercise of the warrants; and |
• |
any other specific terms, preferences, rights or limitations of or restrictions on the warrants. |
59
Before exercising their warrants, holders of warrants will not have any of the rights of holders of the securities purchasable upon such exercise, including:
• |
in the case of warrants to purchase debt securities, the right to receive payments of principal of, or premium, if any, or interest on, the debt securities purchasable upon exercise or to enforce covenants in the applicable indenture; or |
• |
in the case of warrants to purchase common stock or preferred stock, the right to receive dividends, if any, or, payments upon our liquidation, dissolution or winding up or to exercise voting rights, if any. |
Exercise of Warrants
Each warrant will entitle the holder to purchase the securities that we specify in the applicable prospectus supplement at the exercise price that we describe in the applicable prospectus supplement. The warrants may be exercised as set forth in the prospectus supplement relating to the warrants offered. Unless we otherwise specify in the applicable prospectus supplement, warrants may be exercised at any time up to the close of business on the expiration date set forth in the prospectus supplement relating to the warrants offered thereby. After the close of business on the expiration date, unexercised warrants will become void.
Upon receipt of payment and the warrant or warrant certificate, as applicable, properly completed and duly executed at the corporate trust office of the warrant agent, if any, or any other office, including ours, indicated in the prospectus supplement, we will, as soon as practicable, issue and deliver the securities purchasable upon such exercise. If less than all of the warrants (or the warrants represented by such warrant certificate) are exercised, a new warrant or a new warrant certificate, as applicable, will be issued for the remaining warrants.
Governing Law
Unless we otherwise specify in the applicable prospectus supplement, the warrants and any warrant agreements will be governed by and construed in accordance with the laws of the State of New York.
Enforceability of Rights by Holders of Warrants
Each warrant agent, if any, will act solely as our agent under the applicable warrant agreement and will not assume any obligation or relationship of agency or trust with any holder of any warrant. A single bank or trust company may act as warrant agent for more than one issue of warrants. A warrant agent will have no duty or responsibility in case of any default by us under the applicable warrant agreement or warrant, including any duty or responsibility to initiate any proceedings at law or otherwise, or to make any demand upon us. Any holder of a warrant may, without the consent of the related warrant agent or the holder of any other warrant, enforce by appropriate legal action its right to exercise, and receive the securities purchasable upon exercise of, its warrants.
60
We can issue securities in registered form or in the form of one or more global securities. We describe global securities in greater detail below. We refer to those persons who have securities registered in their own names on the books that we or any applicable trustee, depositary or warrant agent maintain for this purpose as the “holders” of those securities. These persons are the legal holders of the securities. We refer to those persons who, indirectly through others, own beneficial interests in securities that are not registered in their own names, as “indirect holders” of those securities. As we discuss below, indirect holders are not legal holders, and investors in securities issued in book-entry form or in street name will be indirect holders.
Book-Entry Holders
We may issue securities in book-entry form only, as we will specify in the applicable prospectus supplement. This means securities may be represented by one or more global securities registered in the name of a financial institution that holds them as depositary on behalf of other financial institutions that participate in the depositary’s book-entry system. These participating institutions, which are referred to as participants, in turn, hold beneficial interests in the securities on behalf of themselves or their customers.
Only the person in whose name a security is registered is recognized as the holder of that security. Securities issued in global form will be registered in the name of the depositary or its participants. Consequently, for securities issued in global form, we will recognize only the depositary as the holder of the securities, and we will make all payments on the securities to the depositary. The depositary passes along the payments it receives to its participants, which in turn pass the payments along to their customers who are the beneficial owners. The depositary and its participants do so under agreements they have made with one another or with their customers; they are not obligated to do so under the terms of the securities.
As a result, investors in a global security will not own securities directly. Instead, they will own beneficial interests in a global security, through a bank, broker or other financial institution that participates in the depositary’s book-entry system or holds an interest through a participant. As long as the securities are issued in global form, investors will be indirect holders, and not legal holders, of the securities.
Street Name Holders
We may terminate a global security or issue securities in non-global form. In these cases, investors may choose to hold their securities in their own names or in “street name.” Securities held by an investor in street name would be registered in the name of a bank, broker or other financial institution that the investor chooses, and the investor would hold only a beneficial interest in those securities through an account he or she maintains at that institution.
For securities held in street name, we or any applicable trustee or depositary will recognize only the intermediary banks, brokers and other financial institutions in whose names the securities are registered as the holders of those securities, and we or any applicable trustee or depositary will make all payments on those securities to them. These institutions pass along the payments they receive to their customers who are the beneficial owners, but only because they agree to do so in their customer agreements or because they are legally required to do so. Investors who hold securities in street name will be indirect holders, not holders, of those securities.
Legal Holders
Our obligations, as well as the obligations of any applicable trustee and of any third parties employed by us or a trustee, run only to the legal holders of the securities. We do not have obligations to investors who hold beneficial interests in global securities, in street name or by any other indirect means. This will be the case whether an investor chooses to be an indirect holder of a security or has no choice because we are issuing the securities only in global form.
For example, once we make a payment or give a notice to the legal holder, we have no further responsibility for the payment or notice even if that legal holder is required, under agreements with its participants or customers or by law, to pass it along to the indirect holders but does not do so. Similarly, we may want to obtain the approval of the legal holders to amend an indenture, to relieve us of the consequences of a default or of our obligation to comply with a particular provision of the indenture or for other purposes. In such an event, we would seek approval only from the holders, and not the indirect holders, of the securities. Whether and how the legal holders contact the indirect holders is up to the legal holders.
61
Special Considerations For Indirect Holders
If you hold securities through a bank, broker or other financial institution, either in book-entry form because the securities are represented by one or more global securities or in street name, you should check with your own institution to find out:
• |
how it handles securities payments and notices; |
• |
whether it imposes fees or charges; |
• |
how it would handle a request for the holders’ consent, if ever required; |
• |
whether and how you can instruct it to send you securities registered in your own name so you can be a holder, if that is permitted in the future; |
• |
how it would exercise rights under the securities if there were a default or other event triggering the need for holders to act to protect their interests; and |
• |
if the securities are in book-entry form, how the depositary’s rules and procedures will affect these matters. |
Global Securities
A global security is a security that represents one or any other number of individual securities held by a depositary. Generally, all securities represented by the same global securities will have the same terms.
Each security issued in book-entry form will be represented by a global security that we issue to, deposit with and register in the name of a financial institution or its nominee that we select. The financial institution that we select for this purpose is called the depositary. Unless we specify otherwise in the applicable prospectus supplement, DTC will be the depositary for all securities issued in book-entry form.
A global security may not be transferred to or registered in the name of anyone other than the depositary, its nominee or a successor depositary, unless special termination situations arise. We describe those situations below under “Special Situations When a Global Security Will Be Terminated.” As a result of these arrangements, the depositary, or its nominee, will be the sole registered owner and legal holder of all securities represented by a global security, and investors will be permitted to own only beneficial interests in a global security. Beneficial interests must be held by means of an account with a broker, bank or other financial institution that in turn has an account with the depositary or with another institution that does. Thus, an investor whose security is represented by a global security will not be a legal holder of the security, but only an indirect holder of a beneficial interest in the global security.
If the prospectus supplement for a particular security indicates that the security will be issued in global form only, then the security will be represented by a global security at all times unless and until the global security is terminated. If termination occurs, we may issue the securities through another book-entry clearing system or decide that the securities may no longer be held through any book-entry clearing system.
Special Considerations for Global Securities
The rights of an indirect holder relating to a global security will be governed by the account rules of the investor’s financial institution and of the depositary, as well as general laws relating to securities transfers. We do not recognize an indirect holder as a holder of securities and instead deal only with the depositary that holds the global security.
If securities are issued only in the form of a global security, an investor should be aware of the following:
• |
an investor cannot cause the securities to be registered in his or her name, and cannot obtain non-global certificates for his or her interest in the securities, except in the special situations we describe below; |
• |
an investor will be an indirect holder and must look to his or her own bank, broker or other financial institution for payments on the securities and protection of his or her legal rights relating to the securities, as we describe above; |
• |
an investor may not be able to sell interests in the securities to some insurance companies and to other institutions that are required by law to own their securities in non-book-entry form; |
• |
an investor may not be able to pledge his or her interest in a global security in circumstances where certificates representing the securities must be delivered to the lender or other beneficiary of the pledge in order for the pledge to be effective; |
62
• |
the depositary’s policies, which may change from time to time, will govern payments, transfers, exchanges and other matters relating to an investor’s interest in a global security; |
• |
we and any applicable trustee have no responsibility for any aspect of the depositary’s actions or for its records of ownership interests in a global security, nor do we or any applicable trustee supervise the depositary in any way; |
• |
the depositary may, and we understand that DTC will, require that those who purchase and sell interests in a global security within its book-entry system use immediately available funds, and your bank, broker or other financial institution may require you to do so as well; and |
• |
financial institutions that participate in the depositary’s book-entry system, and through which an investor holds its interest in a global security, may also have their own policies affecting payments, notices and other matters relating to the securities. |
There may be more than one financial intermediary in the chain of ownership for an investor. We do not monitor and are not responsible for the actions of any of those intermediaries.
Special Situations when a Global Security will be Terminated
In a few special situations described below, the global security will terminate and interests in it will be exchanged for physical certificates representing those interests. After that exchange, the choice of whether to hold securities directly or in street name will be up to the investor. Investors must consult their own banks, brokers or other financial institutions to find out how to have their interests in securities transferred to their own name, so that they will be direct holders. We have described the rights of holders and street name investors above.
Unless we provide otherwise in the applicable prospectus supplement, the global security will terminate when the following special situations occur:
• |
if the depositary notifies us that it is unwilling, unable or no longer qualified to continue as depositary for that global security and we do not appoint another institution to act as depositary within 90 days; |
• |
if we notify any applicable trustee that we wish to terminate that global security; or |
• |
if an event of default has occurred with regard to securities represented by that global security and has not been cured or waived. |
The applicable prospectus supplement may also list additional situations for terminating a global security that would apply only to the particular series of securities covered by the applicable prospectus supplement. When a global security terminates, the depositary, and not we or any applicable trustee, is responsible for deciding the names of the institutions that will be the initial direct holders.
63
The shares of Common Stock being offered by the selling stockholders are those issuable to the selling stockholders, upon exercise of Series A Warrants and Series B Warrants. For additional information regarding the issuances of those shares of common stock and the Series A/B Warrants, see “Private Placement of Common Shares and Warrants” above. We are registering the common stock underlying the Series A/B Warrants in order to permit the selling stockholders to offer the shares for resale from time to time. Except for the ownership of the shares of common stock and the Series A/B Warrants, the selling stockholders have not had any material relationship with us within the past three years.
The table below lists the selling stockholders and other information regarding the beneficial ownership of the shares of common stock by each of the selling stockholders. The second column lists the number of shares of common stock beneficially owned by each selling stockholder, based on its ownership of the shares of common stock and the warrants, as of January 29, 2021, assuming exercise of the Series A/B Warrants held by the selling stockholders on that date, without regard to any limitations on exercises. The third column lists the shares of common stock being offered by this prospectus by the selling stockholders.
In accordance with the terms of the waiver agreements, this prospectus generally covers the resale of at least the maximum number of shares of common stock issued and issuable upon exercise of the Warrants, determined as if the outstanding Warrants were exercised, as applicable, in full, as of the trading day immediately preceding the date this registration statement is initially filed with the SEC. The fourth column assumes the sale of all of the shares offered by the selling shareholders pursuant to this prospectus.
Under the terms of the Series A/B Warrants, a selling stockholder may not exercise the warrants to the extent such exercise would cause such selling stockholder, together with its affiliates, to beneficially own a number of shares of common stock which would exceed 4.99% or 9.99%, as applicable, of our then outstanding common stock following such exercise, excluding for purposes of such determination common stock issuable upon exercise of the warrants which have not been exercised. In addition, pursuant to an agreement with the Company, a selling stockholder cannot receive shares of common stock held in an escrow account for the benefit of such selling stockholder (the “Escrow Common Stock”) to the extent the receipt of such shares would cause such selling stockholder, together with its affiliates, to beneficially own a number of shares of common stock which would exceed 9.99% of the outstanding shares of our then outstanding common stock following such exercise. The number of shares in the second and fourth columns do not reflect this limitation. The selling stockholders may sell all, some or none of their shares in this offering. See “Plan of Distribution.”
Name of Selling Securityholder |
| |
Number of
Shares of
Common
Stock Owned
Prior to
Offering(1)
|
| |
Maximum
Number of
Shares of
Common
Stock to be
Sold
Pursuant to
this
Prospectus
|
| |
Number of
Shares of
Common
Stock Owned
After
Offering
|
| |
Percentage of
Shares of
Common
Stock Owned
After
Offering if
Greater than
1%
|
Empery Asset Master, Ltd.(2)
|
| | 367,875 |
| | 244,560 |
| | 123,315 |
| | *% |
Empery Tax Efficient, LP(3)
|
| | 105,105 |
| | 69,875 |
| | 35,230 |
| | *% |
Empery Debt Opportunity Fund, LP(4)
|
| | 4,782,307 |
| | 3,179,265 |
| | 1,603,042 |
| | 9.99% |
Altium Growth Fund, LP(5)
|
| | 5,295,310 |
| | 3,493,700 |
| | 1,801,610 |
| | 9.9% |
* |
Represents less than 1% |
(1) |
Beneficial ownership includes shares of common stock as to which a person or group has sole or shared voting power or dispositive power. Shares of common stock registered hereunder, as well as shares of common stock subject to options, warrants or convertible preferred stock that are exercisable or convertible within 60 days of January 29, 2021, are deemed outstanding for purposes of computing the number of shares beneficially owned and percentage ownership of the person or group holding such shares of common stock, options, warrants or convertible securities, but are not deemed outstanding for computing the percentage of any other person. |
(2) |
The number of shares consists of (i) 16,561 shares of common stock held directly by the selling stockholder and 106,754 shares of Escrow Common Stock, and (ii) 367,875 shares of common stock issuable upon exercise of the Series A/B Warrants, without giving effect to the blocker provisions described above. Empery Asset Management LP, the authorized agent of Empery Asset Master Ltd (“EAM”), has discretionary authority to vote and dispose of the shares held by EAM and may be deemed to be the beneficial owner of these shares. Martin Hoe and Ryan Lane, in their capacity as investment managers of Empery Asset Management LP, may also be deemed to have investment discretion and voting power over the shares held by EAM. EAM, Mr. Hoe and Mr. Lane each disclaim any beneficial ownership of these shares. |
64
(3) |
The number of shares consists of (i) 4,730 shares of common stock directly by the selling stockholder and 30,500 shares of Escrow Common Stock, and (ii) 69,875 shares of common stock issuable upon exercise of the Series A/B Warrants, without giving effect to the blocker provisions described above. Empery Asset Management LP, the authorized agent of Empery Tax Efficient, LP (“ETE”), has discretionary authority to vote and dispose of the shares held by ETE and may be deemed to be the beneficial owner of these shares. Martin Hoe and Ryan Lane, in their capacity as investment managers of Empery Asset Management LP, may also be deemed to have investment discretion and voting power over the shares held by ETE. ETE, Mr. Hoe and Mr. Lane each disclaim any beneficial ownership of these shares. |
(4) |
The number of shares consists of (i) 215,247 shares of common stock held directly by the selling stockholder and 1,387,795 shares of Escrow Common Stock, and (ii) 3,179,265 shares of common stock issuable upon exercise of the Series A/B Warrants, without giving effect to the blocker provisions described above. Empery Asset Management LP, the authorized agent of Empery Debt Opportunity Fund, LP (“EDOF”), has discretionary authority to vote and dispose of the shares held by EDOF and may be deemed to be the beneficial owner of these shares. Martin Hoe and Ryan Lane, in their capacity as investment managers of Empery Asset Management LP, may also be deemed to have investment discretion and voting power over the shares held by EDOF. EDOF, Mr. Hoe and Mr. Lane each disclaim any beneficial ownership of these shares. |
(5) |
The number of shares consists of (i) 878,561 shares of Common Stock held directly by the selling stockholder and 923,049 shares held in escrow for the benefit of the selling stockholder, and (ii) 3,493,700 shares of Common Stock issuable upon exercise of the Series A/B Warrants, without giving effect to the blocker provision described above. Altium Capital Management, LP, the investment manager of Altium Growth Fund, LP, has voting and investment power over these securities. Jacob Gottlieb is the managing member of Altium Capital Growth GP, LLC, which is the general partner of Altium Growth Fund, LP. Each of Altium Growth Fund, LP and Jacob Gottlieb disclaims beneficial ownership over these shares. |
65
We may sell our securities covered by this prospectus in any of three ways (or in any combination):
• |
to or through underwriters or dealers; |
• |
directly to one or more purchasers; or |
• |
through agents. |
The selling stockholders may sell our securities covered by this prospectus in any of the following ways:
• |
ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
• |
block trades in which the broker-dealer will attempt to sell the securities as agent but may position and resell a portion of the block as principal to facilitate the transaction; |
• |
purchases by a broker-dealer as principal and resale by the broker-dealer for its account; |
• |
an exchange distribution in accordance with the rules of the applicable exchange; |
• |
privately negotiated transactions; |
• |
settlement of short sales; |
• |
in transactions through broker-dealers that agree with the Selling Stockholders to sell a specified number of such securities at a stipulated price per security; |
• |
through the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise; |
• |
on any national securities exchange or quotation service on which the securities may be listed or quoted at the time of sale; |
• |
in the over-the-counter market; |
• |
a combination of any such methods of sale; or |
• |
any other method permitted pursuant to applicable law. |
The selling stockholders may also sell securities under Rule 144 or any other exemption from registration under the Securities Act of 1933, as amended (the “Securities Act”), if available, rather than under this prospectus.
We or the selling stockholders may distribute the securities:
• |
from time to time in one or more transactions at a fixed price or prices, which may be changed from time to time; |
• |
at market prices prevailing at the time of sale; |
• |
at prices related to the prevailing market prices; or |
• |
at negotiated prices. |
Each time we or the selling stockholders offer and sell securities covered by this prospectus, we will provide a prospectus supplement or supplements, if necessary, that will describe the method of distribution and set forth the terms of the offering, including:
• |
the name or names of any underwriters, dealers or agents; |
• |
the amounts of securities underwritten or purchased by each of them; |
• |
the purchase price of securities and the proceeds, if any, we or the selling stockholders will receive from the sale; |
• |
any over-allotment options under which underwriters may purchase additional securities from us or the selling stockholders; |
• |
any underwriting discounts or commissions or agency fees and other items constituting underwriters’ or agents’ compensation; |
66
• |
the public offering price of the securities; |
• |
any discounts, commissions or concessions allowed or reallowed or paid to dealers; and |
• |
any securities exchange or market on which the securities may be listed. |
Any public offering price and any discounts or concessions allowed or reallowed or paid to dealers may be changed from time to time. We or the selling stockholders may determine the price or other terms of the securities offered under this prospectus by use of an electronic auction. We will describe how any auction will determine the price or any other terms, how potential investors may participate in the auction and the nature of the obligations of the underwriter, dealer or agent in the applicable prospectus supplement. In connection with sales of the shares of common stock or otherwise, the selling stockholders may enter into hedging transactions with broker-dealers, which may in turn engage in short sales of the shares of common stock in the course of hedging in positions they assume.
Underwriters or dealers may offer and sell the offered securities from time to time in one or more transactions, including negotiated transactions, at a fixed public offering price or at varying prices determined at the time of sale. If underwriters or dealers are used in the sale of any securities, the securities will be acquired by the underwriters or dealers for their own account and may be resold from time to time in one or more transactions described above. The securities may be either offered to the public through underwriting syndicates represented by managing underwriters, or directly by underwriters or dealers. Generally, the underwriters’ or dealers’ obligations to purchase the securities will be subject to certain conditions precedent. The underwriters or dealers will be obligated to purchase all of the securities if they purchase any of the securities, unless otherwise specified in the prospectus supplement. We may use underwriters with whom we have a material relationship. We will describe the nature of any such relationship in the prospectus supplement, naming the underwriter.
We or the selling stockholders may sell the securities through agents from time to time. The prospectus supplement will name any agent involved in the offer or sale of the securities and any commissions we or the selling stockholders pay to them. Generally, any agent will be acting on a best efforts basis for the period of its appointment. We or the selling stockholders may authorize underwriters, dealers or agents to solicit offers by certain purchasers to purchase the securities from us or the selling stockholders at the public offering price set forth in the prospectus supplement pursuant to delayed delivery contracts providing for payment and delivery on a specified date in the future. The contracts will be subject only to those conditions set forth in the prospectus supplement, and the prospectus supplement will set forth any commissions we or the selling stockholders pay for solicitation of these contracts.
Agents, dealers and underwriters may be entitled to indemnification by us or the selling stockholders against certain civil liabilities, including liabilities under the Securities Act, or to contribution with respect to payments which the agents, dealers or underwriters may be required to make in respect thereof. Agents, dealers and underwriters may be customers of, engage in transactions with, or perform services for us or the selling stockholders in the ordinary course of business.
All securities we may offer, other than common stock, will be new issues of securities with no established trading market. Any underwriters may make a market in these securities, but will not be obligated to do so and may discontinue any market making at any time without notice. We cannot guarantee the liquidity of the trading markets for any securities.
Any underwriter may engage in overallotment, stabilizing transactions, short covering transactions and penalty bids in accordance with Regulation M under the Securities Exchange Act of 1934, as amended. Overallotment involves sales in excess of the offering size, which create a short position. This short sales position may involve either “covered” short sales or “naked” short sales. Covered short sales are short sales made in an amount not greater than the underwriters’ over-allotment option to purchase additional securities in this offering described above. The underwriters may close out any covered short position either by exercising their over-allotment option or by purchasing securities in the open market. To determine how they will close the covered short position, the underwriters will consider, among other things, the price of securities available for purchase in the open market, as compared to the price at which they may purchase securities through the over-allotment option. Naked short sales are short sales in excess of the over-allotment option. The underwriters must close out any naked short position by purchasing securities in the open market. A naked short position is more likely to be created if the underwriters are concerned that, in the open market after pricing, there may be downward pressure on the price of the securities that could adversely affect investors who purchase securities in this offering. Stabilizing transactions permit bids to
67
purchase the underlying security for the purpose of fixing the price of the security so long as the stabilizing bids do not exceed a specified maximum. Penalty bids permit the underwriters to reclaim a selling concession from a dealer when the securities originally sold by the dealer are purchased in a covering transaction to cover short positions.
Any underwriters who are qualified market makers on the Nasdaq Capital Market may engage in passive market making transactions in our common stock, preferred stock, warrants and debt securities, as applicable, on the Nasdaq Capital Market in accordance with Rule 103 of Regulation M, during the business day prior to the pricing of the offering, before the commencement of offers or sales of the securities. Passive market makers must comply with applicable volume and price limitations and must be identified as passive market makers. In general, a passive market maker must display its bid at a price not in excess of the highest independent bid for such security; if all independent bids are lowered below the passive market maker’s bid, however, the passive market maker’s bid must then be lowered when certain purchase limits are exceeded.
The selling stockholders and any broker-dealer participating in the distribution of the shares of common stock may be deemed to be “underwriters” within the meaning of the Securities Act, and any commission paid, or any discounts or concessions allowed to, any such broker-dealer may be deemed to be underwriting commissions or discounts under the Securities Act. At the time a particular offering of the shares of common stock is made, a prospectus supplement, if required, will be distributed which will set forth the aggregate amount of shares of common stock being offered and the terms of the offering, including the name or names of any broker-dealers or agents, any discounts, commissions and other terms constituting compensation from the selling stockholders and any discounts, commissions or concessions allowed or reallowed or paid to broker-dealers.
Under the securities laws of some states, the shares of common stock may be sold in such states only through registered or licensed brokers or dealers. In addition, in some states the shares of common stock may not be sold unless such shares have been registered or qualified for sale in such state or an exemption from registration or qualification is available and is complied with.
There can be no assurance that any selling stockholder will sell any or all of the shares of common stock registered pursuant to the registration statement, of which this prospectus forms a part.
The selling stockholders may pledge or grant a security interest in some or all of the Warrants or shares of common stock owned by them and, if they default in the performance of their secured obligations, the pledgees or secured parties may offer and sell the shares of common stock from time to time pursuant to this prospectus or any amendment to this prospectus under Rule 424(b)(3) or other applicable provision of the Securities Act, amending, if necessary, the list of selling stockholders to include the pledgee, transferee or other successors in interest as selling stockholders under this prospectus. The selling stockholders also may transfer and donate the shares of common stock in other circumstances in which case the transferees, donees, pledgees or other successors in interest will be the selling beneficial owners for purposes of this prospectus.
In compliance with guidelines of the Financial Industry Regulatory Authority, or FINRA, the maximum consideration or discount to be received by any FINRA member or independent broker dealer may not exceed 8% of the aggregate amount of the securities offered pursuant to this prospectus and any applicable prospectus supplement.
Similar to other purchase transactions, an underwriter’s purchase to cover the syndicate short sales or to stabilize the market price of our securities may have the effect of raising or maintaining the market price of our securities or preventing or mitigating a decline in the market price of our securities. As a result, the price of our securities may be higher than the price that might otherwise exist in the open market. The imposition of a penalty bid might also have an effect on the price of the securities if it discourages resales of the securities.
Neither we nor the underwriters make any representation or prediction as to the effect that the transactions described above may have on the price of the securities. If such transactions are commenced, they may be discontinued without notice at any time.
We will pay all expenses of the registration of the shares of common stock pursuant to the waiver agreements, including, without limitation, Securities and Exchange Commission filing fees and expenses of compliance with state securities or “blue sky” laws; provided, however, that a selling stockholder will pay all underwriting discounts and selling commissions, if any. We will indemnify the selling stockholders against liabilities, including some liabilities under the Securities Act, in accordance with the waiver agreements, or the selling stockholders will be entitled to
68
contribution. We may be indemnified by the selling stockholders against civil liabilities, including liabilities under the Securities Act, that may arise from any written information furnished to us by the selling stockholder specifically for use in this prospectus, in accordance with the related waiver agreements, or we may be entitled to contribution.
Once sold under the registration statement, of which this prospectus forms a part, the shares of common stock will be freely tradable in the hands of persons other than our affiliates.
69
Unless otherwise indicated in the applicable prospectus supplement, the validity of the securities offered by this prospectus, and any supplement thereto, will be passed upon for us by Honigman LLP.
Rexahn Pharmaceuticals, Inc. (Effective November 5, 2020 now Ocuphire Pharma, Inc.)
The financial statements of Rexahn Pharmaceuticals, Inc. (effective November 5, 2020 now Ocuphire Pharma, Inc.) as of and for each of the years ended December 31, 2019 and 2018, incorporated in this prospectus by reference to the Annual Report on Form 10-K for the year ended December 31, 2019, have been audited by Baker Tilly US, LLP (formerly known as Baker Tilly Virchow Krause, LLP), an independent registered public accounting firm, as stated in its report thereon incorporated by reference herein, and have been so incorporated in reliance upon such report and upon the authority of such firm as experts in accounting and auditing.
Ocuphire Pharma, Inc.
The financial statements of Ocuphire Pharma, Inc. at December 31, 2019 and 2018, and for each of the two years in the period ended December 31, 2019, incorporated by reference in this Prospectus and Registration Statement have been audited by Ernst & Young LLP, independent registered public accounting firm, as set forth in their report thereon (which contains an explanatory paragraph describing conditions that raise substantial doubt about Ocuphire Pharma, Inc.’s ability to continue as a going concern as described in Note 1 to the financial statements), and are incorporated herein by reference. Such financial statements are, and audited financial statements to be included in subsequently filed documents will be, incorporated herein in reliance upon such report given on the authority of such firm as experts in accounting and auditing.
This prospectus is part of a registration statement we filed with the SEC. This prospectus does not contain all of the information set forth in the registration statement and the exhibits to the registration statement. For further information with respect to us and the securities we are offering under this prospectus, we refer you to the registration statement and the exhibits and schedules filed as a part of the registration statement. Neither we nor any agent, underwriter or dealer has authorized any person to provide you with different information. We are not making an offer of these securities in any state where the offer is not permitted. You should not assume that the information in this prospectus is accurate as of any date other than the date on the front page of this prospectus, regardless of the time of delivery of this prospectus or any sale of the securities offered by this prospectus.
We file annual, quarterly and current reports, proxy statements and other information with the SEC. The SEC maintains a website that contains reports, proxy and information statements and other information regarding issuers that file electronically with the SEC, including Ocuphire. The address of the SEC website is www.sec.gov.
We maintain a website at www.ocuphire.com. Information contained in or accessible through our website does not constitute a part of this prospectus.
70
The SEC allows us to “incorporate by reference” the information we file with it, which means that we can disclose important information to you by referring you to those documents instead of having to repeat the information in this prospectus. The information incorporated by reference is considered to be part of this prospectus, and later information that we file with the SEC will automatically update and supersede this information. We incorporate by reference the documents listed below and any future filings (including those made after the date of the initial filing of the registration statement of which this prospectus is a part and prior to the effectiveness of such registration statement) we will make with the SEC under Sections 13(a), 13(c), 14, or 15(d) of the Exchange Act until the termination of the offering of the shares covered by this prospectus (other than information furnished under Item 2.02 or Item 7.01 of Form 8-K):
• |
Rexahn's annual report on Form 10-K for the year ended December 31, 2019, filed with the SEC on February 21, 2020, as amended on April 29, 2020; |
• |
Rexahn's quarterly reports on Form 10-Q for the quarter ended March 31, 2020, filed with the SEC on May 7, 2020, quarterly report on Form 10-Q for the quarter ended June 30, 2020, filed with the SEC on August 14, 2020, and quarterly report on Form 10-Q for the quarter ended September 30, 2020, filed with the SEC on October 29, 2020; |
• |
Current reports on Form 8-K filed with the SEC on March 16, 2020, April 9, 2020, June 19, 2020, July 1, 2020, August 3, 2020, September 2, 2020, September 11, 2020, October 9, 2020, November 3, 2020, November 6, 2020 (as amended on December 30, 2020), November 17, 2020, November 25, 2020 and February 4, 2021; |
• |
Registration Statement on Form S-4 (File No. 333-239702) filed on July 6, 2020, as amended on August 27, 2020, September 16, 2020 and September 30, 2020; and |
• |
the description of common stock set forth in the Registration Statement on Form 8-A, filed with the SEC on June 7, 2019, including any amendments thereto or reports filed for the purposes of updating this description. |
We will furnish without charge to each person, including any beneficial owner, to whom a prospectus is delivered, upon written or oral request, a copy of any or all of the documents incorporated by reference, including exhibits to these documents.:
Ocuphire Pharma, Inc.
37000 Grand River Avenue, Suite 120
Farmington Hills, MI 48335
(248) 681-9815
Attn: Chief Executive Officer
71
